10-K: Annual report [Section 13 and 15(d), not S-K Item 405]
Published on February 27, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 10-K
For the fiscal year ended December 31 , 2025
or
For the transition period from __________ to __________
Commission file number: 001-41968
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) | (IRS Employer Identification No.) | |||||||
| (Address of Principal Executive Offices) | (Zip Code) | |||||||
(Registrant’s Telephone Number, Including Area Code) ( | ||||||||
| Securities registered pursuant to Section 12(b) of the Act: | ||||||||||||||
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐
Indicate by checkmark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of "large accelerated filer," "accelerated filer," "smaller reporting company," and "emerging growth company" in Rule 12b-2 of the Exchange Act:
| ☒ | Accelerated filer | ☐ | |||||||||
| Non-accelerated filer | ☐ | Smaller reporting company | |||||||||
| Emerging growth company | |||||||||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by checkmark whether the registrant has filed a report on and attestation to its management's assessment of the effectiveness of its internal controls over financial reporting under Section 404(b) of the Sarbanes Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by checkmark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant's executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
The aggregate market value of the outstanding common stock of the Registrant held by non-affiliates as the last business day of the registrant's most recently completed second fiscal quarter, was approximately $13.1 billion. There were 173,493,005 shares of common stock with a par value of $0.01 per share outstanding as of February 18, 2026.
DOCUMENTS INCORPORATED BY REFERENCE
Parts of the Company's definitive proxy statement (to be filed pursuant to Regulation 14A within 120 days after the Registrant's fiscal year-end of December 31, 2025) for its annual meeting to be held on May 15, 2026, are incorporated by reference in this Form 10-K in response to Part III, Items 10, 11, 12, 13 and 14.
1
SOLVENTUM CORPORATION
FORM 10-K
For the Year Ended December 31, 2025
TABLE OF CONTENTS | PAGE | ||||
2
TABLE OF CONTENTS | PAGE | ||||
3
Cautionary Note Concerning Forward Looking Statements
This Annual Report on Form 10-K, including "Management’s Discussion and Analysis of Financial Condition and Results of Operations" in Part II, Item 7, and other materials Solventum has filed or will file with the SEC (and oral communications that Solventum may make) contain or incorporates by reference statements that relate to future events and expectations and, as such, constitute forward-looking statements that involve risk and uncertainties. Forward-looking statements include those containing such words as "anticipates," "believes," "could," "estimates," "expects," "forecasts," "goal," "guidance," "intends," "may," "outlook," "plans," "projects," "seeks," "sees," "should," "targets," "will," "would," or other words of similar meaning.
All statements that reflect Solventum’s expectations, assumptions or projections about the future, other than statements of historical fact, are forward-looking statements, including, without limitation, forecasts relating to discussions of future operations and financial performance (including volume growth, pricing, sales and earnings per share growth and cash flows) and statements regarding Solventum’s strategy for growth, future product development, regulatory clearances and approvals, competitive position and expenditures. Forward-looking statements are not guarantees of future performance and are subject to risks, uncertainties, and changes in circumstances that are difficult to predict, including, but not limited to, the factors described in Part I, Item 1A, "Risk Factors" in this Annual Report on Form 10-K, which are summarized below. Although Solventum believes that the expectations reflected in any forward-looking statements it makes are based on reasonable assumptions, it can give no assurance that these expectations will be attained and it is possible that actual results may differ materially from those indicated by these forward-looking statements due to a variety of risks and uncertainties. Forward-looking statements are based on certain assumptions and expectations of future events and trends, and actual future results and trends may differ materially from historical results or those reflected in any such forward-looking statements depending on a variety of factors. Solventum assumes no obligation to update or revise such statement, whether as a result of new information, future events or otherwise, except as required by applicable law.
Summary of Risk Factors
Solventum’s business is subject to numerous risks and uncertainties that could adversely affect Solventum’s business, results of operations, financial condition and cash flows. These risks include, but are not limited to, the following, all of which are more fully described in Part I, Item 1A "Risk Factors", and should be considered an integral part of Part II, Item 7, "Management’s Discussion and Analysis of Financial Condition and Results of Operations." This summary should be read in conjunction with the Risk Factors section and should not be relied upon as an exhaustive summary of the material risks facing Solventum’s business.
•Solventum’s historical financial information for periods prior to the Spin-Off is not necessarily representative of the results or performance that it would have achieved as a separate, publicly traded company.
•Solventum may not achieve some or all of the expected benefits of the Spin-Off.
•Solventum’s accounting and other management systems and resources may not be adequately prepared to meet the financial reporting and other requirements to which it is subject to as a standalone publicly traded company.
•In connection with the Spin-Off, Solventum incurred debt obligations and may incur additional obligations in the future, which could adversely affect its business and profitability and its ability to meet other obligations.
•Solventum may not be able to engage in desirable capital-raising or strategic transactions following the Spin-Off.
•If the Spin-Off, together with certain related transactions, were to fail to qualify as a transaction that is generally tax-free for U.S. federal income tax purposes, Solventum and its shareholders could be subject to significant tax liabilities.
•The transfer to Solventum of certain contracts, permits and other assets and rights may have required the consents or approvals of, or provide other rights to, third parties and governmental authorities.
•Following the Spin-Off, Solventum’s commercial relationships with 3M remain significant, which could adversely affect Solventum’s business, its ability to meet other obligations and the market price of its common stock.
•Solventum’s results may be impacted by the effects of, and changes in, worldwide economic, political, regulatory, international trade and geopolitical conditions, war and other events beyond its control.
•The deployment of artificial intelligence or other emerging technologies in Solventum's products and services, or a failure to adapt its products and services, could affect future results.
•Public health crises may increase Solventum’s cost of doing business and disrupt Solventum’s operations.
•Our brands are critical to our success, and damage to our reputation or our brands could adversely affect our business, results of operations or financial condition.
•Acquisitions, strategic alliances, divestitures and other strategic events resulting from portfolio management actions and other evolving business strategies, and possible further organizational restructuring, could affect future results.
•Solventum may not be able to effectively integrate acquired businesses into its operations or achieve expected cost savings or profitability from its acquisitions.
•Solventum's restructuring program may not be successful.
•Solventum may not be able to access the capital and credit markets on terms that are favorable to Solventum, or at all.
•Change in Solventum’s credit ratings could increase cost of funding.
4
•Changes in foreign currency exchange rates or interest rates could adversely affect Solventum.
•Solventum operates in highly competitive markets, competition may increase in the future and the healthcare industry may be disrupted, necessitating that Solventum lower prices or resulting in a loss of market share.
•Consolidation in the healthcare industry could have an adverse effect on Solventum’s revenues and results of operations.
•Reductions in customers’ research budgets or government funding may adversely affect Solventum’s business.
•Solventum’s growth objectives are largely dependent on the timing and market acceptance of its new products and services.
•The success of many of Solventum’s products depends upon certain key healthcare professionals.
•Changes in reimbursement practices of third-party payers or other cost containment measures or worsening economic conditions could affect the demand for Solventum’s products and the prices at which they are sold.
•Solventum’s future results are subject to vulnerability with respect to materials and availability of purchased components, compounds, raw materials, energy, production capacity and labor.
•3M is the sole source of supply for raw materials used in certain of our products and our business will be harmed if 3M does not satisfy our requirements.
•Solventum is subject to risks related to international, federal, state and local treaties, laws and regulations, as well as related compliance risks.
•Solventum may face potential liabilities related to PFAS, which could adversely impact Solventum’s results.
•The impacts of climate change may adversely effect Solventum’s business.
•Solventum operates in a strictly regulated industry, and compliance with laws and regulations applicable to the commercialization of Solventum’s products is costly and failure to comply may result in significant penalties.
•Solventum is subject to laws and regulations governing government contracts and public procurement in many jurisdictions, as to which the failure to comply could adversely affect Solventum’s business.
•Solventum is exposed to risks associated with product liability claims, including existing claims and claims resulting from the actions or inactions of its customers or third parties, and product recalls or safety alerts that are outside of its control.
•Security and data breaches, cyberattacks and other cybersecurity incidents involving Solventum’s information technology systems and infrastructure could disrupt or interfere with Solventum’s operations.
•Solventum may be unable to obtain, maintain, protect or effectively enforce its intellectual property rights.
•Changes in tax rates, laws or regulations could adversely impact Solventum’s financial results.
•Solventum’s tax burden could increase as a result of ongoing or future tax audits and inquiries.
•Solventum could be negatively impacted by future changes in the allocation of income to each of the income tax jurisdictions in which Solventum operates.
•If Solventum is unable to attract or retain key personnel and qualified employees, or maintain relations with its employees, unions and other employee representatives, Solventum’s business would be adversely affected.
•A significant number of shares of Solventum common stock may be sold by 3M or others, which may cause the Solventum stock price to decline.
•Because Solventum does not currently intend to pay any dividends on its common stock, holders of its common stock must rely on stock appreciation for any return on their investment.
•Anti-takeover provisions could enable Solventum’s Board of Directors to resist a takeover attempt by a third party and limit the power of its shareholders.
5
SOLVENTUM CORPORATION
ANNUAL REPORT ON FORM 10-K
For the Year Ended December 31, 2025
PART I
Item 1. Business
Solventum Corporation ("Solventum," "we," "our," "us," or the "Company"), is a leading global healthcare company developing, manufacturing, and commercializing a broad portfolio of solutions that leverages deep material science, data science, and digital capabilities to address critical customer and patient needs. We constantly seek to enable the improvement of standards of care and move healthcare forward with innovation powered by insights, clinical intelligence, technology, and manufacturing expertise. Our 70+ year history of discovering and innovating advanced solutions has helped us solve our customers’ toughest challenges.
Our solutions are relied on every day within the global healthcare industry to deliver higher-quality patient care, more efficient processes and workflows, and improved standards of safety and accuracy. Additionally, our products and services are present along a patient’s journey through prevention, diagnosis, treatment, and recovery.
Our business possesses strong customer relationships, a broad, wide-ranging, and well-known portfolio of brands, differentiated technology, and manufacturing expertise. We serve a diverse customer base, ranging from multidisciplinary hospitals to local clinics/practices. Our long-tenured and collaborative customer relationships globally give us unique insights into their needs and preferences. These insights inform our innovation processes, drive stronger customer retention, and create multiple avenues for further customer engagement.
Business Segments
We are organized into three reportable operating business segments that are aligned with the markets we serve.
MedSurg (57.9% of 2025 total sales) is a provider of solutions including negative pressure wound therapy, advanced wound dressings, advanced skin care, synthetic tissue matrices, I.V. site management, sterilization assurance, temperature management, surgical supplies, medical tapes and wraps, stethoscopes, medical electrodes, and medical technologies Original Equipment Manufacturer ("OEM"). These solutions are designed to accelerate healing, prevent complications, and lower the total cost of care. Additionally, our comprehensive range of surgical solutions are designed to mitigate a patient’s risk of infection or complications.
Dental Solutions (16.2% of 2025 total sales) is a provider of a comprehensive suite of dental and orthodontic products including brackets, aligners, restorative cements, and bonding agents that span the "life of the tooth," including products designed for preventative dental care, direct and indirect restoration, and broad orthodontic needs.
Health Information Systems (16.3% of 2025 total sales) provides healthcare systems with software solutions – including computer-assisted physician documentation, direct-to-bill and coding automation, classification methodologies, speech recognition, and data visualization platforms – that are designed to eliminate revenue cycle waste, create more time for patient care, and support value-based care. These solutions are designed to ensure accuracy of reimbursement and reduce the administrative burden that clinicians face.
Acquisitions and Divestitures
As part of our business strategy, Solventum intends to monitor its business portfolio and organizational structure and may make acquisitions and divestitures that expand or enhance its organizational structure.
In 2025, the Company acquired Acera Surgical ("Acera"), a privately held bioscience company. Also in 2025, the Company sold its Purification and Filtration business to Thermo Fischer Scientific Inc. Refer to Note 3, "Acquisitions and Divestitures" for additional information on acquisitions and divestiture activity in periods presented in this Annual Report on Form 10-K.
Research and Development Activities
Our Research and Development ("R&D") activities are focused on developing new solutions that are clinically supported and differentiated as well as improving on our marketed solutions to address evolving customer needs and enable better outcomes and access for patients. Our R&D capabilities include R&D organizations that operate within each of our business segments, as well as R&D capabilities spanning across our business segments.
Our business segment R&D organizations are responsible for the full product development life cycle, leveraging industry insights, domain-specific expertise in end-to-end product development, and a detailed understanding of customer applications
6
and usability to innovate in both new and marketed products. Our cross-segment capabilities include building new shared technologies and advancing existing shared technologies. We believe that collaboration across our organization further enhances our R&D capabilities by standardization of common processes and sharing of best practices, enabling consistent and collaborative development and issue resolution, promoting synergies in development and manufacturing, and creating a broad culture of solving what matters.
Our R&D team consists of approximately 2,000 employees, including research scientists, chemical engineers, data scientists, software engineers, and product developers. They are supported by a team of accomplished clinicians from our medical affairs. We partner with our medical and scientific affairs to enhance our clinical insight and expand awareness of clinical studies regarding our solutions by increasing both the number of peer-reviewed publications and the visibility for existing publications that address our solutions.
Intellectual Property
Development and protection of our proprietary technologies through IP rights is a strategic priority for our business. To protect our proprietary technologies, Solventum relies on a combination of patent, design, utility model, trademark, copyright, and trade secret protections as well as regulatory exclusivity periods and confidentiality agreements. Our IP team collaborates with our R&D and product teams to develop product line focused IP strategies and secure IP rights as appropriate. We generally file patent applications in the United States and foreign countries that have strong technology patent protections. We also license from third parties IP that complements our internal R&D efforts and product offerings. While, in aggregate, our patents and other IP are vital to our operations, we do not consider any single IP asset or group of assets to be of material importance to any segment or to the business as a whole; rather, we believe understanding our customers’ needs, technology expertise, and manufacturing know-how are critical for our business.
Competitors
We operate in highly competitive markets across our segments and product categories throughout the world. Our ability to compete effectively is contingent upon several factors, including but not limited to our ability to deliver differentiated clinical and economic outcomes for our customers.
In the MedSurg segment, the advanced wound care market is highly competitive, particularly in the United States and Europe, with our principal competitors including Smith & Nephew, Medaxis, Mölnlycke, Coloplast, and Convatec. Our infection prevention and surgical supplies solutions are offered in highly competitive and fragmented end markets, especially in the United States and Europe. Our principal competitors in this segment include Becton Dickinson, Hartmann, ICU Medical, Medline, Cardinal Health, Fortive, Steris, MDF Instruments, and BSN.
The dental market is highly competitive, with players ranging from very large broad-based multinational companies to localized or specialized suppliers and start-ups. Principal multinational competitors within the oral care market include Dentsply Sirona, Envista, and Straumann, all of which compete with both dental and orthodontic solutions, and Ivoclar, which competes with only dental solution offerings, and Align Technology, which competes with only orthodontic solution offerings.
The health care software technology market in which we conduct our business, and the healthcare information technology ("HCIT") industry in general, is highly competitive and dynamic, characterized by the continual introduction of new products and technologies. Principal competitors include Optum, Microsoft (Nuance), Epic, Oracle (Cerner), Waystar, Athena, and a host of start-up technologies actively working to disrupt the areas of revenue cycle management and clinician productivity. In the United States, the market for value-based care software solutions is highly fragmented and subject to continuous entry of new competitors. The market is even more fragmented internationally. Outside the U.S., we compete primarily with local players in the respective country or region, in addition to new market entrants.
Human Capital
We have a long-tenured and diverse talent base with significant work experience, technical qualifications, and healthcare industry expertise. Our employee base consists of approximately 20,000 employees, with approximately 40% having more than 10 years of tenure. We have approximately 10,000 employees in the United States and approximately 2,000 in Germany. Of our employees, approximately 4,000 are production employees working in plants across the globe. We do not have any unionized facilities in the US. Our relationship with employee-representative organizations outside the U.S. takes many forms, including in European Union countries where we engage with representative bodies for employees, such as employee forums, works councils and trade unions, in accordance with local law.
Our employees are united in our mission to provide better, smarter, safer healthcare to improve lives. Our culture highlights collaboration and teamwork, along with a focus on empathy, solving challenges and improving care.
7
The ability to recruit, retain, develop, protect, and fairly compensate our global workforce will be a key driver of our success. Our key human capital priorities are designed to support those efforts, including those listed below.
•Health and Safety: Solventum is committed to the safety, health, and well-being of its employees. We continuously evaluate opportunities to raise safety and health standards, visiting sites to identify and manage environmental health and safety risks; evaluating compliance with regulatory requirements and company policies; and maintaining a global security operation for the protection of facilities and people on our sites. We also promote health and well-being through disease prevention programs, on-site clinical services, employee assistance programs, and comprehensive healthcare benefits.
•Development and Pipeline: We have a robust, continuous talent review process focused on succession planning and key talent development. We deploy functional and leadership development opportunities and support time to learn. In addition, we advance our talent pipeline by attracting competitive talent and accelerating leadership and key skill development. We are expanding our external talent pools and leveraging university and professional organizations to identify talent with critical skills.
• Workplace Environment: Our ability to evolve as a highly preferred organization who serves the needs of our employees and contributes to customer success is realized by enabling a workplace and environment which fosters belonging. In support of the communities we operate in globally, we are committed to inclusion in its broadest sense. We strengthen our commitment by ensuring accessible and fair processes for all. Whether we are attracting or advancing talent, we focus on conveying our intent for leaders and employees to thrive. Engaging talent such as those who are involved in our Employee Impact Groups, which are open to everyone, has proven to be a way to empower and inspire our workforce as they drive innovation in unique ways.
•Compensation and Benefits: Our total compensation for employees includes a variety of components that support sustainable employment and the ability to build a strong financial future, including competitive market-based pay and comprehensive benefits. In addition, we have a professional and flexible work environment that promotes innovation and well-being and rewards performance.
Environmental, Health, and Safety Matters
Solventum benefits from having both an Environmental Health and Safety (EHS) organization and a Product Stewardship (PS) organization which collaborate to ensure environmental, health and safety ("EHS") considerations are proactively managed across facilities and products. We are subject to various laws, regulations, ordinances, customer requirements, and industry standards related to EHS matters. These include, but are not limited to, permitting, licensing, and authorization requirements, and regulatory obligations. These affect a significant portion of our activities globally across each of our segments and product lines and require compliance related to, among other things: (a) occupational health, safety, and well-being; (b) the protection of the environment; (c) emissions and discharges to air and water; (d) greenhouse gas management and climate change; (e) the use of natural resources; (f) the handling, use, storage, transportation, and disposal of toxic or hazardous materials, radioactive materials, and solid and hazardous wastes; and (g) the procurement and use of select materials and chemicals. EHS laws and regulations also vary widely by jurisdiction, may be established at supranational, international, national, state, and/or local levels, and are constantly evolving, often to become more stringent.
Compliance with EHS laws, regulations, customer requirements, and industry standards require, among other things, that we maintain and operate our equipment safely; obtain and keep current environmental permits, radioactive material licenses, and radiation machine registrations; install pollution control technologies; and maintain certain records and submit specific reports. Failure to comply could lead to enforcement actions, such as the imposition of civil or criminal fines and penalties; the suspension or termination of our permits, licenses, authorizations, or operations; claims by third parties; remediation expenses or liabilities; or other sanctions.
Solventum also is subject to a broad and continually evolving set of regulations governing the manufacture, processing, distribution, import, export, and labeling of its products and their raw materials. Examples include various chemical control regulations, such as European Union ("EU") Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), and global variations, which require registration, evaluation including potential testing, reporting and possible restrictions. In addition, Solventum complies with global Extended Producer Responsibility (EPR) reporting requirements for products containing electronics and batteries; electrical safety regulations; Globally Harmonized System of Classification and Labeling of Chemicals (GHS); and various other chemical, material, product, and packaging requirements including communication and reporting obligations.
Beyond safety and compliance, Solventum practices responsible chemical use, working to minimize hazards wherever possible. As an example, a specific chemical category of interest is Per - and polyfluoroalkyl substances ("PFAS"). A small number of Solventum’s products utilize and/or contain PFAS. Use is critically evaluated on an on-going basis, and non-PFAS alternatives
8
are utilized when possible. Regulatory and legislative activities concerning PFAS are accelerating in the United States, Europe and elsewhere. Solventum diligently tracks and analyzes these global emerging regulations for applicability to both products and operations, taking a proactive approach and ensuring preparedness.
Solventum’s operations are also subject to various laws governing occupational health and safety. An example in the US is the federal Occupational Safety and Health Act ("OSHA") and parallel state and local occupational health and safety standards. These standards establish certain employer responsibilities, including requirements to maintain a workplace free of recognized hazards likely to cause serious injury or death, certain medical and hygiene standards, licensing and permitting obligations and various recordkeeping, disclosure and procedural requirements. Solventum’s facilities and operations may be subject to periodic inspections by OSHA representatives and comparable authorities in other jurisdictions. Failure to comply with applicable occupational health and safety standards, even if no work-related serious injury or death occurs, could result in civil or criminal enforcement and substantial penalties, significant capital expenditures or suspension or limitation of Solventum’s operations.
In addition, Solventum manufacturing facilities are also subject to environmental, health and safety statutes, regulations and permit requirements, including, but not limited to, applicable requirements under the Clean Air Act, the Clean Water Act, the Resource Conservation and Recovery Act, the Seveso-III Directive (Directive 2012/18/EU), the European Machinery Directive (Directive 2006/42/EC), the EU Industrial Emissions Directive (Directive 2010/75/EU), and the European Pollutant Release and Transfer Register.
Sales and Marketing
We have an extensive global commercial footprint with sales in over 90 countries. To serve our diverse customer base across our prioritized geographies, we take a multi-model commercial approach, including direct-to-customer, distribution, key account management, inside sales, and e-commerce. We augment our commercial model with both marketing and service support. Key marketing activities include brand management, insights, price management, digital marketing, and integrated marketing communications. As our customers increasingly leverage both traditional and digital media in their path to purchase, we continue to optimize our own omnichannel execution to ensure the best customer experience possible. Our service support teams include clinical specialists (licensed nurses or technicians), medical liaisons (clinical professionals such as surgeons and dentists), and application engineers (technical subject matter experts). These teams provide high-quality customer support serving as the clinical and/or technical expert for the customer.
To expand our market coverage into emerging geographies in the Latin America, Europe, Middle East and Africa, and Asia regions, we employ an export commercial model and leverage local partners to market and sell our products. Our local third-party partners facilitate regulatory and cross-border import compliance and distribute directly to the customer. Our export commercial team provides technical, clinical, and marketing support both directly to our customers as well as to our third-party partners, to help increase customer satisfaction and support our ability to grow our global presence.
Global Supply Chain and Sourcing
Our Global Supply Chain strategy is designed to ensure speed, reliability, and cost efficiency, with the flexibility and resilience to navigate market changes.
Our sourcing, production, and distribution network is managed globally, supported by advanced manufacturing and assembly capabilities across our worldwide footprint. Our manufacturing is supported by a global distribution network. Our sourcing practices prioritize sustainable suppliers and comply with global regulatory standards.
Our distribution network is strategically designed using a "hub-and-spoke" model. This approach optimizes route planning and accelerates delivery times to customers across all regions. This footprint is "purpose-built" to meet customer needs in a cost-effective manner.
As we separate from 3M, we continue to leverage 3M's established infrastructure across certain geographies ensuring continuity and reliability during our transition, while we invest in expanding our independent capabilities. The use of both distribution networks and manufacturing sites is covered under a Transition and Distribution Agreement and a Transition Contract Manufacturing Agreement, respectively, with 3M.
Our supply chain resiliency program consisting of regional sources of supply, dual-source manufacturing capabilities and vertically integrated operations presents a competitive advantage by providing a reliable supply of products to our customers.
In 2025, the United States government announced new tariffs on imported goods from certain countries, and in response, foreign governments have threatened or imposed tariffs or other actions. On February 20, 2026, the United States Supreme Court issued a decision concluding that the International Emergency Economic Powers Act does not provide authority for the
9
President to impose tariffs. During 2025, certain tariffs that affected us were imposed under this statute pursuant to presidential executive order. The ultimate financial impact of this decision cannot be reasonably estimated at this time. The extent and timing of any potential recoveries of tariffs previously paid remain subject to further legal interpretation and administrative processes. We will continue to monitor developments and will evaluate the effect of the ruling on future reporting periods as additional information becomes available.
Health Care Policies
Political, economic, technological and regulatory influences around the world continue to subject the health care industry to potential fundamental changes that could substantially affect our results of operations. We maintain a global Government Affairs presence to actively monitor and advocate on a myriad of legislation and policies that may potentially impact us, both on a domestic and an international front. The Government Affairs office works closely with members of Congress and committee staff, the White House and administration offices, state governors, legislatures and regulatory agencies, embassies and global governments on issues affecting our business. The Government Affairs office also advocates for public policy that benefits our employees and the patients we serve and supports the communities in which we live.
Our products are purchased principally by hospitals, physicians and other health care providers around the world that typically bill various third-party payers, including government programs (e.g., Medicare and Medicaid in the U.S.) and private insurance payers, for the items and services provided to their patients. Government and private sector initiatives related to limiting the growth of health care costs (including price regulation), coverage and payment policies, comparative effectiveness reviews of therapies, technology assessments, price transparency, health care delivery and payment structure reforms and localization or regionalization of supply, are continuing in many countries where we do business. We believe that these changes are causing the marketplace to place increased emphasis on the delivery of treatments that can reduce costs, improve efficiencies and/or increase patient access. Implementation of cost containment initiatives and health care reforms in significant markets such as the U.S., China and other markets may limit the price of, or the level at which reimbursement is provided for, our products or procedures using our products, which in turn may make it less likely that a hospital or physician will select our products to treat patients.
Regulations
General
The regulations applicable to Solventum are promulgated and enforced by government bodies in individual countries and govern the methods and controls used for the design, manufacture, packaging labeling, storage, safety, sales and distribution, marketing clearance or approval, advertising and promotion, sterilization, installation, servicing, performance and effectiveness of the products Solventum sells globally. In addition, Solventum is subject to other complex and varying regulations related to anti-trust, anti-corruption, export control, privacy and data protection, emerging technologies, such as AI, and other areas.
These regulations are dynamic and
•apply to all facilities of Solventum’s business that conduct the foregoing activities, regardless of where the facilities are located;
•apply to the activities performed by most of Solventum’s employees, including but not limited to sales and marketing, research and development, regulatory affairs, quality assurance, medical affairs, and operations, both before and after a product is commercially distributed; and
•differ by country and/or region.
Solventum commits a significant amount of resources to maintain compliance with these regulations, to monitor evolving or new regulations, to respond to requests, inspections, and/or audits from governing bodies that monitor compliance with relevant regulations, and develop products that adhere to regulatory requirements in order to obtain regulatory clearances and/or approvals. Compliance with these regulations requires Solventum to create systems, processes, and procedures that are aligned with the regulations in all markets Solventum serves. Compliance also requires Solventum to maintain knowledge of the current regulations that govern its activities. As these regulations change, Solventum must adapt its systems, processes, and procedures to comply with the new regulations.
See "Item 1A. Risk Factors" for a description of the risks related to the regulations that are applicable to Solventum.
Regulation of Medical Devices and Pharmaceutical Products
The products Solventum develops, manufactures, and commercializes are regulated in most of the markets Solventum serves. Some of these products meet the definition of medical devices, pharmaceuticals or combination products and are regulated, as such, by various governmental bodies, globally. All products produced by the MedSurg and Dental Solutions segments, with
10
very few exceptions, meet the definition of a medical device or pharmaceutical product. Accordingly, the development, manufacture, and commercialization of these products must comply with the regulations governing medical devices or pharmaceuticals in the markets we serve. Conversely, none of the products produced by the Health Information Systems segment meet the definition of medical devices or pharmaceuticals. The product portfolio of the segment is dynamic and changes with time, depending on the needs of the customers served. While the Health Information Systems segment does not currently include medical devices or pharmaceutical products, this may change in the future.
Determinations of the safety and efficacy of our products are solely within the authority of the United States Food and Drug Administration ("FDA") or other applicable regulatory authorities in jurisdictions outside the United States.
United States
In the United States, the Food, Drug, and Cosmetic Act authorizes the Food and Drug Administration to oversee and regulate the production, sale, and distribution of food, drugs, medical devices, and cosmetics.
Medical Devices
Solventum’s medical devices are regulated by the FDA’s Center for Devices and Radiological Health. The FDA classifies medical devices into one of three classes depending on the degree of risk associated with the medical device and the extent of manufacturer and regulatory control needed to ensure its safety and effectiveness: Class I (Low Risk), Class II (Moderate Risk), and Class III (High Risk).
•Class I: Class I medical devices are those where the General Controls for Medical Devices are sufficient to provide a reasonable assurance of safety and effectiveness.
•Class II: Class II medical devices are where the General Controls for Medical Devices alone are insufficient to provide reasonable assurance of its safety and effectiveness and there is sufficient information to establish special controls, including the promulgation of performance standards, post-market surveillance, patient registries, development and dissemination of guidelines, recommendations, and other appropriate actions as the FDA Commissioner deems necessary to provide such assurance.
•Class III: Class III medical devices are intended to be used in supporting or sustaining human life or preventing impairment of human health, or that may present a potential unreasonable risk of illness or injury for which the General Controls for Medical Devices and special controls are insufficient to provide reasonable assurance of the safety and effectiveness of a device, or for which there is insufficient information to make such a determination. Class III devices typically require pre-market approval.
Most medical devices are cleared by the FDA through the Pre-Market Notification, or 510(k), process. This process requires medical device manufacturers to demonstrate that their devices are as safe and effective as (i.e., substantially equivalent to) a legally marketed medical device. Pre-market notifications are required for most Class II and some Class I medical devices. Due to the level of risk associated with Class III devices, the FDA has determined that these devices require a pre-market approval (a "PMA"). A PMA application is the most stringent type of marketing application required by the FDA. Typically, PMA submissions require the submission of human clinical trials to achieve FDA approval. Currently, none of the medical devices in Solventum’s portfolio of products are Class III medical devices in the United States.
Pharmaceutical Products
Solventum’s pharmaceutical products are regulated by the FDA’s Center for Drug Evaluation and Research. Generally, drugs are brought to the market through the New Drug Application ("NDA") process or an Over-the-Counter ("OTC") Monograph. Solventum’s pharmaceutical products are brought to market through the NDA and OTC pathways.
The NDA process typically begins with the completion of extensive preclinical laboratory tests and preclinical animal studies, which may need to be performed in accordance with the Good Laboratory Practices regulations, followed by the submission to the FDA of an investigational new drug application which must become effective before human clinical trials may begin and must be updated annually.
Before each clinical study may be initiated, an independent Institutional Review Board or ethics committee representing each clinical site must approve such study. The clinical studies must be adequate, well-controlled and conducted in accordance with Good Clinical Practice ("GCP") requirements.
After the completion of the clinical trials, an NDA is submitted to the FDA to demonstrate that a drug is safe and effective in its proposed use(s), the benefits of the drug outweigh the risks, the drug’s proposed labeling (package insert) is appropriate, and
11
the methods used to manufacture the drug are adequate to preserve the drug’s identity, strength, quality, and purity. Following the completion of the clinical trials, the NDA process with the FDA generally involves the following:
•preparation of and submission to the FDA of an NDA;
•potential review of the product application by an FDA advisory committee, where appropriate and if applicable;
•a determination by the FDA within 60 days of its receipt of an NDA to file the application for review;
•satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities where the proposed product drug substance and drug product are produced to assess compliance with current Good Manufacturing Processes ("cGMP"), and audits of selected clinical trial sites to ensure compliance with GCP; and
•FDA review and approval of an NDA prior to any commercial marketing or sale of the drug or biologic in the United States.
An OTC drug monograph is a type of "recipe book" covering acceptable ingredients, doses, formulations, labeling, and, in some cases, testing parameters. OTC drug monographs are continually updated to add additional ingredients and labeling as needed. Products conforming to a monograph may be marketed without FDA pre-approval. Under the OTC monograph system, selected OTC drugs are generally recognized as safe and effective and do not require the submission and approval of an NDA. The FDA OTC monographs include well-known ingredients and specific requirements for permitted indications, required warnings and precautions, allowable combinations of ingredients and dosage levels. Pharmaceutical products marketed under the OTC monograph system must conform to specific quality, formula and labeling requirements. Facilities where OTC drugs are manufactured, tested, packaged, stored or distributed must comply with cGMP regulations and/or regulations promulgated by the FDA or other competent authorities. OTC monograph products that do not comply with these standards can be deemed unapproved new drugs and can be required to be withdrawn from the market.
European Union
Medical Devices
All medical devices that are placed on the market or put into service in the European Union must meet the requirements of the Medical Device Regulation. Manufacturers must demonstrate compliance to the requirements of the Medical Device Regulation, prior to affixing the CE Mark on the products and commercializing in the European Union. A CE Mark is a symbol placed on a product that declares that the product is compliant with the essential requirements of applicable health, safety and environmental protection regulation. Compliance to the Medical Device Regulation requires a manufacturer to demonstrate that its products comply with minimum standards of performance, safety, and quality, through a conformity assessment procedure that depends on the product’s classification. The Medical Device Regulation describes four classes of medical devices: Class I (Lowest Risk), Class IIa, Class IIb, and Class III (Highest Risk). Classification is dependent on a variety of factors, including duration of use, whether the device is invasive or non-invasive, and whether the device is considered "active." Notified bodies are responsible for ensuring that manufacturers comply with the requirements of the Medical Device Regulation.
Pharmaceutical Products
The European Medicines Agency is responsible for regulating pharmaceutical products in the European Union. The European Medicines Agency implements a system similar to the U.S. FDA’s Center for Drug Evaluation and Research and it is responsible for evaluating the quality, safety, and efficacy of drug products in the EU. If the European Medical Agency concludes that all requirements for efficacy, safety and quality are met, it issues a positive opinion that is forwarded to the European Commission and the European Commission makes the final decision on the granting of a marketing authorization.
China
The chief pharmaceutical product and medical device regulator in China is the National Medical Products Administration ("NMPA"), which enforces medical device and pharmaceutical product laws and regulations and standards and has the power to issue fines, seize products, withdraw or suspend an approval or a registration for serious non-compliances, and refer cases for criminal prosecution. These national laws and regulations are also supplemented by provincial and other local-level rules and enforcement policies.
Medical Devices
Locally manufactured medical devices gain market authorization through municipal authorities, while medical devices that are not manufactured in China are reviewed by the NMPA and must be accompanied by appropriate documentation showing that the device has been approved in its country of origin.
12
Medical devices are classified into three classes: Class I (Lowest Risk), Class II, and Class III (Highest Risk). Approved products are subject to post-market requirements for reporting adverse events and recalls, as well as regular risk assessments of devices and potentially re-evaluation reports of the safety and effectiveness of the device based on more significant safety signals.
In addition to product licenses, manufacturing and distribution facilities that handle Class II and III devices require licenses or notifications and must comply with cGMP requirements and good supply practices. The NMPA regularly conducts inspections of manufacturing facilities in China (as part of a pre-market submission review, routine or for-cause inspections, or unannounced inspections) as well as periodic inspections of overseas manufacturers for compliance with China medical device cGMP requirements. The NMPA inspects distributors and user facilities and conducts annual national and provincial sampling inspections and testing to ensure compliance with labeling, licensing, mandatory standards, and other related requirements. In addition, the NMPA conducts regular and for-cause good clinical practice audits of clinical sites that provide data and clinical trial reports for product registration.
Pharmaceutical Products
Solventum’s pharmaceutical products are strictly regulated by the NMPA and various provincial, city, and county regulators. Some of Solventum’s pharmaceutical products require pre-market approval from the NMPA before they can be marketed in China, and those marketing applications must be supported by clinical data, which typically comes from a multi-phase study in China or by relying on clinical data generated abroad that meets the NMPA’s requirements.
Regulation on Advertising, Marketing, and Promotion
The advertising, marketing, and promotion of Solventum’s products must be truthful and non-misleading, consistent with applicable regulatory clearances and approvals, and supported by adequate and reasonable scientific data. Solventum typically is required to have a reasonable basis to support any factual marketing claims, and what constitutes a reasonable basis for substantiation can vary widely from market to market and from product to product.
With limited exceptions, Solventum may not market, promote, or sell regulated products prior to regulatory authority clearance or approval. Regulatory authorities, including the FDA and other government bodies in the jurisdictions where Solventum markets its products, strictly regulate the indications for use and associated promotional safety and effectiveness claims that may be made about approved or cleared products.
Data Privacy Laws
Solventum is also subject to extensive laws and regulations protecting the privacy, security, and integrity of personal information, including patient medical information, that it receives, including, among others, the U.S. Health Insurance Portability and Accountability Act of 1996, as amended, (the "HIPAA"), the California Consumer Privacy Act (the "CCPA"), and similar U.S. state laws, the European Union’s General Data Protection Regulation (the "E.U. GDPR"), the United Kingdom’s Data Protection Act 2018 (the "UK DPA") and the General Data Protection Regulation (the "U.K. GDPR"), and China’s Personal Information Protection Law ("PIPL"), and Personal Data Cross Border Transfer Rule ("CBDT"). Federal health information privacy laws, such as HIPAA, and consumer protection laws impose requirements for the collection, use, storage, access, transfer and protection of health-related and other sensitive and personal information, and failure to comply may result, such as with respect to any CCPA violations, in civil penalties. The CCPA has been amended by the California Privacy Rights Act ("CPRA"), which came into effect, in most material respects, on January 1, 2023, and was most recently updated again in 2025. The most recent updates address cybersecurity audits, risk assessments, and automated decision-making technology. The E.U. GDPR and the U.K. GDPR and UK DPA, together with national legislation, regulations and guidelines of the E.U. Member States and the United Kingdom governing the processing of personal data, impose strict obligations and restrictions on the ability to collect, analyze, store, transfer and otherwise process personal data, including health data and adverse event reporting. The E.U. GDPR contemplates fines for certain violations of up to four percent of global annual revenue or €20 million (or GBP 17.5 million under the U.K. GDPR), whichever is greater, and enforcement can include limits on data transfers to countries outside the E.U. or U.K. In China, Solventum is subject to PIPL, which applies to the secure processing of personal information of natural persons within China, and Solventum is also subject to CBDT for the processing of personal information outside China where the purpose is to provide products and services within China and the analysis or assessment of the activities of individuals within China. Consequences of non-compliance with PIPL may include monetary fines of up to five percent of the previous year’s revenue, termination of data transfers and personal liability imposed on those directly responsible. Solventum is also subject to similar privacy and data protection frameworks in other developed and emerging markets. While Solventum utilizes industry standard processes, including the National Institute of Standards and Technology (the "NIST") privacy framework and third-party management processes, to assess the potential impact of emerging laws and enforcement trends on its business and to mitigate potential impacts on its business, data privacy laws and regulations and their scope and enforcement are constantly evolving, and Solventum cannot predict what effect, if any, changes to these laws and regulations and Solventum’s compliance with them may have on its business.
13
Global Healthcare Compliance
The marketing, promotion, and sale of medical devices, drugs, and services is regulated by the U.S. Department of Health and Human Services and equivalent U.S. state and non-U.S. agencies responsible for reimbursement and regulation of the delivery of healthcare items and services. These include laws and regulations related to kickbacks, false claims, self-referrals, and healthcare fraud and abuse. Similar regulations are imposed at the state level, as well as in many global markets in which we do business.
The U.S. FCPA, the U.K. Bribery Act of 2010, and similar anti-corruption and anti-bribery laws in other jurisdictions generally prohibit companies from promising to pay money or anything of value to any foreign official for the purpose of obtaining or retaining business. These laws apply to many of Solventum’s customer interactions, as healthcare professionals in other countries are or are often considered government officials, and in some cases lay out specific requirements of how to comply or demonstrate compliance with the legal requirements.
Quality and Safety
The FDA and comparable authorities in other jurisdictions regulate the facilities and operational procedures that Solventum uses to manufacture its products. Solventum is required to register its facilities with these authorities. Solventum’s products are required to be manufactured in facilities that operate in accordance with current cGMPs. The FDA and comparable authorities in other jurisdictions periodically inspect Solventum’s manufacturing facilities for compliance with cGMP or similar manufacturing standards in the applicable country.
Solventum is also required to establish and follow quality systems to comply with certain post-market surveillance requirements, specifically those pertaining to adverse event reporting for medical devices and pharmaceuticals. For medical devices, Solventum is required to report deaths and serious injuries that a device may have caused or to which a device may have contributed. Solventum is also required to report certain device malfunctions. For pharmaceuticals, Solventum is required to report any undesirable event that is associated with the use of a drug or biological product in humans whether or not the events are considered to be product related. For both medical devices and pharmaceuticals, Solventum is required to establish and implement quality systems to comply with regulations pertaining to post-market surveillance.
In addition, many of Solventum’s products are subject to regulation by the Consumer Product Safety Commission (the "CPSC") under the Consumer Product Safety Act and other laws enforced by the CPSC. These statutes and related regulations establish safety standards and bans for consumer products. The CPSC monitors compliance of consumer products under its jurisdiction through market surveillance and has the authority to conduct product safety inspections of establishments where consumer products are manufactured, held or transported. The CPSC can require the recall of noncompliant products or products containing a defect that creates a substantial risk of injury to the public, and the CPSC may seek penalties for regulatory noncompliance under certain circumstances. CPSC regulations also require manufacturers of consumer products to report to the CPSC certain types of information regarding products that fail to comply with applicable regulations, contain a defect that could create a substantial product hazard or create an unreasonable risk of serious injury or death. Certain state laws also address the safety of consumer products and may mandate reporting or labeling requirements.
Pricing
Solventum’s activities are subject to a variety of price control laws, regulations and government mandates in some of the markets in which it operates. In certain markets the pricing for certain of Solventum’s products may be subject to prior approval, including in jurisdictions where Solventum’s products are subject to government reimbursement, whereas in other markets Solventum may be able to set its own prices for its products subject to certain degrees of monitoring and control by the applicable governmental authority.
Environmental, Health, and Safety Laws
Solventum is subject to a broad range of federal, state, provincial and local environmental laws and regulations concerning environmental, health and safety matters. For a discussion of these laws and regulations, see the section titled "Environmental, Health, and Safety Matters."
14
Information about our Executive Officers
Below is a list of the executive officers of the Company and other significant employees who are members of our leadership team. The respective age of each individual in the tables below is as of December 31, 2025. No family relationships exist among any of the executive officers named, nor is there any undisclosed arrangement or understanding pursuant to which any person was selected as an officer. This information is presented in the table below as of the date of the 10-K filing (February 27, 2026).
Name | Age | Position | ||||||||||||
Bryan Hanson | 59 | Chief Executive Officer | ||||||||||||
Wayde McMillan | 56 | Chief Financial Officer | ||||||||||||
| Heather Knight | 54 | Chief Commercial Officer | ||||||||||||
Tammy Gomez | 54 | Chief Human Resources Officer | ||||||||||||
Paul Harrington | 57 | Chief Supply Chain Officer | ||||||||||||
Marcela Kirberger | 59 | Chief Corporate & Legal Affairs Officer | ||||||||||||
Amy Landucci | 51 | Chief Information & Digital Officer | ||||||||||||
The following are brief biographies describing the backgrounds of our executive officers.
Bryan Hanson. Mr. Hanson has served as the Chief Executive Officer of Solventum since 2024 after serving as the Chief Executive Officer of 3M’s Health Care Business Group from September 2023 to the Spin-Off. Mr. Hanson has also served as a director of Solventum since 2024. Previously, Mr. Hanson served as Chairman of the Board of Directors of Zimmer Biomet, a global medical technology company with annual revenues over $7 billion, from May 2021 to August 2023, and as President and Chief Executive Officer and a member of the Board of Zimmer Biomet from December 2017 to August 2023. Previously, Mr. Hanson served as Executive Vice President and President, Minimally Invasive Therapies Group of Medtronic plc, a medical device company, from January 2015 until joining Zimmer Biomet in December 2017. Prior to that, he was Senior Vice President and Group President of Covidien plc, a medical device company, from October 2014 to January 2015; Senior Vice President and Group President, Medical Devices and United States of Covidien from October 2013 to September 2014; Senior Vice President and Group President of Covidien for the Surgical Solutions business from July 2011 to October 2013; and President of Covidien’s Energy-based Devices business from July 2006 to June 2011. Mr. Hanson held several other positions of increasing responsibility in sales, marketing and general management with Covidien from October 1992 to July 2006. Mr. Hanson holds a Bachelor of Science degree in Finance from Florida State University. He also completed the Kellogg School of Management Finance for Executives program in 2010 and the Harvard Executive Education in Leadership program in 2013.
Wayde McMillan. Mr. McMillan has served as the Chief Financial Officer of Solventum since 2024 after serving as the Chief Financial Officer of 3M’s Health Care Business Group since November 2023. Previously, Mr. McMillan served as Executive Vice President, Chief Financial Officer and Treasurer of Insulet, a medical device company, from March 2019 to November 2023. From January 2015 to February 2019, he was Chief Financial Officer and Vice President of Finance of the Minimally Invasive Therapies Group at Medtronic plc., a medical device company. From November 2006 to January 2015, prior to Medtronic’s acquisition of Covidien plc, a medical device company, Mr. McMillan held a variety of leadership positions at Covidien, including Chief Financial Officer and Vice President of Finance of the Medical Devices Group & U.S., Chief Financial Officer and Vice President of Finance of the Surgical Solutions Business Unit, and Vice President Finance and Controller of the Respiratory and Monitoring Solutions Business Unit. Mr. McMillan started his career in accounting, audit, financial analysis and investor relations positions at various institutions. Mr. McMillan earned his Bachelor of Science in Business Administration from Merrimack College and an MBA from Bentley University McCallum Graduate School of Business. Mr. McMillan currently serves on the board of directors of Hologic Inc.
Heather Knight. Ms. Knight has served as Chief Commercial Officer of Solventum since November 2025. Ms. Knight has more than 30 years of experience in the healthcare industry. Prior to joining Solventum, she served as Chief Operating Officer of Baxter International, where she was responsible for global sales across three business segments, as well as research and development, supply chain, and medical and regulatory affairs. From 2019 to 2025, Ms. Knight held several senior leadership positions at Baxter, including interim Group President of its Medical Products and Therapies segment, Regional Division President, and Global Business Unit President for Medication Delivery, Clinical Nutrition and Acute Therapies. Earlier in her career, she held leadership roles in general management, upstream innovation, and commercial strategy at Medtronic plc, Covidien plc, Tyco International plc, and Kendall Healthcare Products Company. Ms. Knight earned her bachelor's degree in Biological Sciences from the University of Buffalo and completed the Executive Sales and Management program from the University of Chicago Booth School of Management. Ms. Knight currently serves on the board of directors of Waters Corporation.
15
Tammy Gomez. Ms. Gomez has served as the Chief Human Resources Officer of Solventum since 2024. Before joining Solventum, Ms. Gomez was Executive Vice President and Chief Human Resource Officer at Owens & Minor from July 2022 to December 2023. After starting as a Corporate Audit Manager in 1999, she spent more than 20 years at Cardinal Health in various roles of increasing responsibility, including most recently leading its Global HR Center of Excellence & HR Services team. She began her career in finance and worked with Prudential and General Motors. Ms. Gomez earned her Bachelor’s degree in Business Administration and Management from the University of South Alabama.
Paul Harrington. Mr. Harrington has served as the Chief Supply Chain Officer of Solventum since 2024. Mr. Harrington spent 10 years at Medtronic, from November 2013 to November 2023, where he was most recently Senior Vice President, Global Operations Innovation responsible for shaping, transforming and innovating across the global operations and supply chain function. He also previously served as Vice President Global Operations for the Restorative Therapies Group and Neuromodulation business unit. Prior to joining Medtronic, he was Vice President of Global Manufacturing at Covidien. Mr. Harrington earned his Bachelor's degree in Business Administration from Kingston College.
Marcela Kirberger. Ms. Kirberger has served as the Chief Corporate and Legal Affairs Officer and Corporate Secretary of Solventum since 2026, and was previously the Chief Legal Affairs Officer since 2024. Previously she served as the Chief Legal Affairs Officer of 3M’s Health Care Business Group since November 2023. Previously, Ms. Kirberger was Executive Vice President, General Counsel and Corporate Secretary at Elanco, a pharmaceutical company in the animal health field, from June 2021 to November 2023, where she was responsible for the global strategy and operations of the legal function and had responsibility for Corporate Affairs, ESG and Enterprise Risk Management programs. Prior to joining Elanco, Ms. Kirberger held U.S., regional and global leadership roles within Fortune 500 life sciences companies, including as General Counsel and Corporate Secretary at Roche Diagnostics NA, a diagnostics company, from 2019 to 2021; and General Counsel and Chief Compliance Officer at Leica Microsystems GmbH, a global medical device manufacturer and subsidiary of Danaher Corporation, from 2017 to 2019. She also worked for the Novartis Group of Companies from 2006 to 2017 in different Novartis companies, including Gerber Products, Novartis Consumer Health, Novartis Pharmaceuticals and Sandoz International, where she was Global Head of Legal for Biopharma and before that, Global Chief Compliance Officer. Ms. Kirberger began her career as a securities litigator at Lowenstein Sandler in New Jersey. A native of Argentina, she earned her law degrees from Rutgers School of Law in Newark, New Jersey and the Catholic University of Argentina in Buenos Aires.
Amy Landucci. Ms. Landucci has served as the Chief Information and Digital Officer of Solventum since 2024. Prior to joining Solventum, Ms. Landucci was Chief Digital and Technology Officer at Haleon from July 2022 until April 2024, where she led the technical carveout from GSK to form a standalone consumer healthcare company. Prior to the carveout, she spent five years from July 2017 until July 2022 at GSK and previously spent a decade at Novartis in a variety of roles. The first eight years of her career were in consulting with Accenture. Ms. Landucci has served on the Board of Directors for Healthy Women, the leading independent, non-profit health information source for women in the U.S. Ms. Landucci earned her Bachelor of Arts in Environmental Studies from Gustavus Adolphus College.
Ethics and Governance
All of Solventum's employees, including its Chief Executive Officer, Chief Financial Officer, Controller and Chief Accounting Officer, are required to abide by Solventum’s Code of Conduct to ensure that its business is conducted in a consistently legal and ethical manner. Solventum’s Code of Conduct covers many topics, including antitrust and competition law, conflicts of interest, financial reporting, protection of confidential information, and compliance with all laws and regulations applicable to the conduct of its business.
Employees are required to report any conduct that they believe in good faith to be an actual or apparent violation of the Code of Conduct. Solventum Legal Affairs team maintains processes to receive, retain, and address concerns received from internal, external or anonymous sources. Solventum employs a team of experienced investigators who review the reported concerns and conduct investigations when necessary. Solventum maintains the confidentiality of reported concerns in accordance with legal requirements and in the best interest of the organization and those involved. Solventum does not tolerate retaliation for anyone who raises a genuine concern in good faith and educates employees on this policy. Information on how to submit any such communications can be found on Solventum’s website, under "Ethics and Compliance." Solventum’s Chief Compliance Officer has a direct reporting obligation to the Audit Committee and regularly reports to the Audit Committee on compliance with Solventum's Code of Conduct, including the effectiveness of Solventum's compliance program. See "Item 10: Directors, Executive Officers and Corporate Governance – Code of Ethics" for information about our Code of Ethics governing our Chief Executive Officer, Chief Financial Officer and Controller and Chief Accounting Officer.
Solventum’s Board of Directors has also implemented a Code of Business Conduct and Ethics for Directors of Solventum. This Code incorporates principles of conduct at Solventum and Solventum’s Board of Directors are to follow to ensure Solventum’s business, and the activities of Solventum’s Board of Directors are conducted with integrity and adherence to the highest ethical standards, and in compliance with the law. Solventum’s Code of Conduct for employees and the Code of Business Conduct and Ethics for Directors are available on Solventum’s website at www.solventum.com.
16
Additional Information
Solventum’s Internet address is www.solventum.com. Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended (the "Exchange Act"), are available on our website for free as soon as reasonably practicable after they are filed electronically with the SEC. Reports filed with the SEC may be viewed at sec.gov. The information on our website is not, and shall not be deemed to be, a part of this Annual Report on Form 10-K or incorporated into any other filings we make with the SEC.
Item 1A. Risk Factors
RISK FACTORS
Investing in our securities involves a high degree of risk. You should consider and read carefully all of the risks and uncertainties described below, as well as the other information contained in this Annual Report, including our financial statements included elsewhere in this Annual Report, before making an investment decision. The risks described below are not the only ones we face. The occurrence of any of the following risks or additional risks and uncertainties not presently known to us or that we currently believe to be immaterial could cause a material adverse effect on our business, results of operations, financial condition and liquidity. In addition, many of these risks are interrelated and could occur under similar business and economic conditions, and the occurrence of certain of them could in turn cause the emergence or exacerbate the effect of others. The risk factors described below are not necessarily presented in order of importance. This Annual Report also contains forward-looking statements and estimates that involve risks and uncertainties. Our actual results could differ materially from those anticipated in the forward-looking statements as a result of specific factors, including the risks and uncertainties described below. See "Special Note Regarding Forward-Looking Statements and Information."
Risks Related to the Spin-Off and Solventum’s Relationship with 3M
Solventum’s historical financial information for periods prior to the Spin-Off is not necessarily representative of the results or performance that it would have achieved as a separate, publicly traded company and may not be a reliable indicator of its future results or performance.
The historical financial information of Solventum for periods prior to the Spin-Off included in this Annual Report is derived from the Consolidated Financial Statements and accounting records of 3M. Accordingly, the financial information included herein does not necessarily reflect the financial condition, results of operations or cash flows that Solventum would have achieved as a separate, publicly traded company during the periods prior to the Spin-Off or those that Solventum will achieve in the future primarily as a result of the factors described below:
•Prior to the Spin-Off, Solventum’s working capital requirements and capital for its general corporate purposes, including capital expenditures and acquisitions, had historically been satisfied as part of the corporate-wide cash management policies of 3M. Following the Spin-Off, Solventum’s results of operations or cash flows may be more vulnerable to changing market conditions and therefore more volatile.
•Prior to the Spin-Off, 3M or one of its affiliates performed various corporate functions for Solventum’s business such as information technology, legal, treasury, accounting, auditing, human resources, investor relations and finance. The financial results included in the historical financial information reflect allocations of corporate expenses from 3M for such functions, which may be less than the expenses that Solventum would have incurred had it operated as a separate, publicly traded company. Solventum may also be unable to replicate corporate functions that will operate with the same degree of effectiveness as the equivalent 3M functions from which the Solventum business had historically benefited.
•Solventum’s business historically was integrated with the other businesses of 3M and benefited from 3M’s economies of scope and scale in costs, employees, vendor relationships and customer relationships. While Solventum has sought to minimize the impact on its business when separating these arrangements, there is no guarantee these arrangements will continue to capture these benefits in the future. Additionally, Solventum may be unable to obtain similar arrangements, goods, services or technologies at prices or on terms as favorable as or to the same extent that 3M obtained them prior to the Spin-Off. Among other benefits, Solventum’s business historically had access to 3M’s extensive global research and development resources, which historically enhanced its ability to innovate, develop new products and technologies, and improve and update existing products and technologies. Solventum’s lack of access to 3M’s research and development resources may negatively impact it.
•The cost of capital for Solventum’s business may be higher than 3M’s cost of capital prior to the Spin-Off, and Solventum may need to obtain additional financing from banks, through public offerings or private placements of debt
17
or equity securities, strategic relationships or other arrangements in order to fund capital expenditures and investments, return capital to shareholders or service debt, which may or may not be available and may be more costly.
•Solventum’s historical financial information for periods prior to the Spin-Off does not reflect the debt that Solventum incurred as part of the Spin-Off.
•Solventum has and will continue to incur costs in connection with the separation from 3M and the related transition agreements entered into with 3M.
•As an independent public company, Solventum is subject to public company reporting requirements under the Exchange Act, and is required to prepare its standalone financial statements according to the rules and regulations required by the SEC.
Other significant changes may occur in Solventum’s cost structure, management, financing and business operations as a result of operating as a company separate from 3M.
Solventum may not achieve some or all of the expected benefits of the Spin-Off.
Solventum may not be able to achieve the full strategic and financial benefits expected to result from the Spin-Off, or such benefits may be delayed or not occur at all. The Spin-Off was designed to provide the following benefits to Solventum, among others: (1) the ability to pursue tailored capital allocation strategies and make company-specific investment decisions to drive innovation and growth; (2) enhanced management focus, with Solventum having a distinct board and management team with relevant expertise able to focus on strengthening its business; (3) improved operational agility and focus, enabling Solventum to pursue its distinct operating priorities and strategies with increased flexibility to act based on its unique characteristics, better positioning each for long-term success; (4) greater access to capital through the creation of a distinct and compelling investment profile appealing to a different long-term investor base from 3M; (5) independent equity currency, enabling Solventum to use its own industry-focused stock to consummate future acquisitions or other transactions; and (6) enhanced recruitment and retention, including by aligning employee, management and board incentives with performance.
Solventum may not achieve these and other anticipated benefits for a variety of reasons, including, among others: (1) the operation of Solventum as a standalone public company continues to demand significant management resources and require significant amounts of management’s time and effort, which may divert management’s attention from operating and growing Solventum’s business; (2) Solventum may continue to be required to pay costs that could be substantial and material to its financial resources, including costs associated with separating its operations from 3M and for related transition services, as well as increased accounting, tax, legal and other professional services costs; and (3) operational challenges as a standalone company, including those related to customer service, pace of change and productivity improvements, could result in additional expenses or reductions in productivity. If Solventum fails to achieve some or all of the benefits expected to result from the Spin-Off, or if such benefits are delayed, it could have a material adverse effect on its competitive position, business, financial condition, results of operations and cash flows.
Solventum’s accounting and other management systems and resources may not be adequately prepared to meet the financial reporting and other requirements to which it is subject to as a standalone publicly traded company following the Spin-Off.
Solventum’s financial results were previously included within the consolidated results of 3M. Solventum was not directly subject to the reporting and other requirements of the Exchange Act. As a result of the Spin-Off, Solventum is directly subject to reporting and other obligations under the Exchange Act, including the requirements of Section 404 of Sarbanes-Oxley Act, which require annual management assessments of the effectiveness of its internal control over financial reporting and a report by its independent registered public accounting firm. These reporting and other obligations place significant demands on Solventum’s management and administrative and operational resources, including accounting resources.
To comply with these requirements, Solventum has needed to migrate its systems, including information technology systems and enterprise resource planning systems, implement additional financial and management controls, reporting systems and procedures and hire additional accounting and finance staff. Solventum has incurred and expects to incur additional annual expenses related to these steps, and those expenses may be significant. If Solventum is unable to implement appropriate financial and management controls, reporting systems, information technology and procedures in a timely and effective fashion, its ability to comply with its financial reporting requirements and other rules that apply to reporting companies under the Exchange Act could be impaired. In addition, implementation of management controls, reporting systems, information technology and procedures may result in additional material costs to Solventum. Any failure to achieve and maintain effective internal controls in a timely, effective and cost-effective manner could have a material adverse effect on its business, financial condition, results of operations and cash flows.
18
In connection with the Spin-Off, Solventum incurred debt obligations and may incur additional obligations in the future, which could adversely affect its business and profitability and its ability to meet other obligations.
In connection with the Spin-Off, Solventum completed certain financing transactions. As a result of such transactions and the subsequent repayment of debt with proceeds from the divestiture of the Purification and Filtration business, as of December 31, 2025, Solventum had approximately $5 billion of outstanding indebtedness. Depending on market or other economic conditions, Solventum could also incur additional indebtedness in the future.
This significant amount of debt could potentially have important consequences to Solventum and its fixed income and equity investors, including:
•requiring a substantial portion of its cash flow from operations to make interest payments;
•making it more difficult to satisfy debt service and other obligations;
•increasing the risk of a future credit ratings downgrade, which could increase future debt costs and limit the future availability of debt financing;
•increasing its vulnerability to general adverse economic and industry conditions;
•reducing the cash flow available to fund capital expenditures and other corporate purposes and to grow its business;
•limiting Solventum’s flexibility in planning for, or reacting to, changes in its business and the industry;
•placing Solventum at a competitive disadvantage relative to its competitors that may not be as highly leveraged with debt;
•requiring Solventum to repatriate earnings to the U.S., causing withholding taxes to be applied, which in turn could increase Solventum’s effective tax rate; and
•limiting Solventum’s ability to raise additional capital or borrow additional funds as needed or take advantage of business opportunities as they arise, pay cash dividends or repurchase ordinary shares.
To the extent that Solventum incurs additional indebtedness, the foregoing risks could increase. In addition, Solventum’s actual cash requirements in the future may be greater than expected. Its cash flow from operations may not be sufficient to repay all of the outstanding debt as it becomes due, and Solventum may not be able to borrow money, sell assets or otherwise raise funds on acceptable terms, or at all, to refinance its debt.
Solventum may not be able to engage in desirable capital-raising or strategic transactions following the Spin-Off.
Under current U.S. federal income tax law, a spin-off that otherwise qualifies for tax-free treatment can be rendered taxable to the parent corporation and its shareholders as a result of certain post-spin-off transactions, including certain acquisitions of shares or assets of the spun-off corporation. To preserve the tax-free treatment of the distribution and certain related transactions, and in addition to Solventum’s indemnity obligation described below, the Tax Matters Agreement restricts Solventum, for the two-year period following the distribution, except in specific circumstances, from (1) entering into any transaction pursuant to which all or a portion of the shares of Solventum stock would be acquired, whether by merger or otherwise; (2) issuing equity securities beyond certain thresholds; (3) repurchasing shares of Solventum stock other than in certain open-market transactions; and (4) ceasing to actively conduct certain of Solventum’s businesses. Further, the Tax Matters Agreement imposes similar restrictions on Solventum and its subsidiaries that are intended to prevent certain transactions undertaken as part of the internal reorganization from failing to qualify as transactions that are generally tax-free for U.S. federal income tax purposes under Sections 355 and 368(a)(1)(D) of the Code or for applicable non-U.S. income tax purposes. The Tax Matters Agreement also prohibits Solventum from taking or failing to take any other action that would prevent the distribution and certain related transactions (or certain transactions undertaken as part of the internal reorganization) from qualifying as tax-free transactions under applicable law. These restrictions may limit Solventum’s ability to pursue certain equity issuances, strategic transactions or other transactions that it may otherwise believe to be in the best interests of its shareholders or that might increase the value of its business.
If the Spin-Off, together with certain related transactions, were to fail to qualify as a transaction that is generally tax-free for U.S. federal income tax purposes, Solventum, as well as its shareholders, could be subject to significant tax liabilities. In addition, if certain internal restructuring transactions were to fail to qualify as transactions that are generally tax-free for U.S. federal or non-U.S. income tax purposes, Solventum could be subject to significant tax liabilities. In certain circumstances, Solventum could be required to indemnify 3M for material taxes and other related amounts pursuant to indemnification obligations under the Tax Matters Agreement.
It was a condition to the Spin-Off that (1) the private letter ruling received by 3M from the U.S. Internal Revenue Service (the "IRS") regarding certain U.S. federal income tax matters relating to the Spin-Off, including the qualification of the Spin-Off, together with certain related transactions, as a transaction that is generally tax-free for U.S. federal income tax purposes pursuant to Sections 355 and 368(a)(1)(D) of the Internal Revenue Code of 1986, as amended (the "Code," such qualification, "U.S. Tax-Free Status" and, such ruling, the "IRS Ruling") be valid; and (2) 3M received one or more opinions of 3M’s tax advisors regarding U.S. Tax-Free Status (each, a "Tax Opinion"). The IRS Ruling is and any Tax Opinion were based upon and
19
rely on, among other things, various facts and assumptions, as well as certain representations, statements and undertakings of 3M and Solventum, including those relating to the past and future conduct of 3M and Solventum. If any of these representations, statements or undertakings are, or become, inaccurate or incomplete, or if any representations or covenants contained in any of the separation-related agreements and documents or in any documents relating to the IRS Ruling and/or any Tax Opinion are inaccurate or not complied with by 3M, Solventum or any of their respective subsidiaries, the IRS Ruling and/or such Tax Opinion may be invalid and the conclusions reached therein could be jeopardized.
Notwithstanding 3M’s receipt of the IRS Ruling and any Tax Opinion, in each case, regarding U.S. Tax-Free Status, the IRS could determine that the distribution and/or certain related transactions should be treated as taxable transactions for U.S. federal income tax purposes if it determines that any of the representations, assumptions or undertakings upon which the IRS Ruling or such Tax Opinion was based are inaccurate or have not been complied with. In addition, the IRS Ruling does not address all of the issues that are relevant to determining whether the Spin-Off, together with certain related transactions, qualifies as a transaction that is generally tax-free for U.S. federal income tax purposes, and any Tax Opinion represents the judgment of such advisor and is not binding on the IRS or any court and the IRS or a court may disagree with the conclusions in such Tax Opinion. Accordingly, notwithstanding 3M’s receipt of the IRS Ruling and the Tax Opinion(s), in each case, regarding U.S. Tax-Free Status, there can be no assurance that the IRS will not assert that the Spin-Off and/or certain related transactions do not qualify for tax-free treatment for U.S. federal income tax purposes, or that a court would not sustain such a challenge. In the event the IRS were to prevail with such challenge, 3M, Solventum and their shareholders could be subject to significant U.S. federal income tax liability.
If the Spin-Off and certain related transactions were to fail to qualify as a transaction that is generally tax-free for U.S. federal income tax purposes under Sections 355 and 368(a)(1)(D) of the Code, in general, for U.S. federal income tax purposes, 3M would recognize a taxable gain as if it had sold the Solventum common stock distributed to 3M shareholders in a taxable sale for its fair market value, and 3M shareholders who received Solventum common stock in the distribution would be subject to tax as if they had received a taxable distribution equal to the fair market value of such shares. Even if the distribution and certain related transactions otherwise qualify as generally tax-free for U.S. federal income tax purposes under Section 355 and Section 368(a)(1)(D) of the Code, it may result in taxable gain to 3M (but not its shareholders) under Section 355(e) of the Code if the distribution were deemed to be part of a plan (or series of related transactions) pursuant to which one or more persons acquire, directly or indirectly, shares representing a 50 percent or greater interest (by vote or value) in 3M or Solventum. For this purpose, any acquisitions of 3M or Solventum shares within the period beginning two years before the Spin-Off and ending two years after the distribution are presumed to be part of such a plan, although 3M or Solventum may be able to rebut that presumption. The process for determining whether an acquisition is part of a plan under these rules is complex, inherently factual in nature and subject to a comprehensive analysis of the facts and circumstances of the particular case. Notwithstanding the IRS Ruling and the Tax Opinion(s), a sufficient change in ownership of 3M or Solventum may occur that could result in a material tax liability to 3M.
In addition, prior to the Spin-Off, 3M and its subsidiaries completed the internal reorganization, and 3M, Solventum and their respective subsidiaries incurred certain tax costs in connection with the internal reorganization, including non-U.S. tax costs resulting from transactions in non-U.S. jurisdictions, which may be material. With respect to certain transactions undertaken as part of the internal reorganization, 3M has requested and intends to obtain tax rulings in certain non-U.S. jurisdictions and/or opinions of external tax advisors, in each case, regarding the tax treatment of such transactions. Such tax rulings and opinions will be based upon and rely on, among other things, various facts and assumptions, as well as certain representations, statements and undertakings of 3M, Solventum or their respective subsidiaries. If any of these representations or statements is, or becomes, inaccurate or incomplete, or if 3M, Solventum or any of their respective subsidiaries do not fulfill or otherwise comply with any such undertakings or covenants, such tax rulings and/or opinions may be invalid or the conclusions reached therein could be jeopardized. Further, notwithstanding receipt of any such tax rulings and/or opinions, there can be no assurance that the relevant taxing authorities will not assert that the tax treatment of the relevant transactions differs from the conclusions reached in the relevant tax rulings and/or opinions. In the event any such tax rulings and/or opinions cannot be obtained or the relevant taxing authorities prevail with any challenge in respect of any relevant transaction, Solventum and 3M could be subject to significant tax liabilities.
Under the Tax Matters Agreement that Solventum entered into with 3M, Solventum generally is required to indemnify 3M for any taxes resulting from the separation (and any related costs and other damages) to the extent such amounts resulted from (1) an acquisition of all or a portion of Solventum’s equity securities or assets, whether by merger or otherwise (and regardless of whether Solventum participated in or otherwise facilitated the acquisition), (2) other actions or failures to act by Solventum or (3) certain of Solventum’s representations, covenants or undertakings being incorrect or violated. Any such indemnity obligations could be material. In addition, Solventum, 3M and each company’s respective subsidiaries may have incurred certain tax costs in connection with the separation, which may be material.
The transfer to Solventum of certain contracts, permits and other assets and rights may have required the consents or approvals of, or provide other rights to, third parties and governmental authorities. If such consents or approvals were not
20
obtained, Solventum may not be entitled to the full benefit of such contracts, permits and other assets and rights, which could increase its expenses or otherwise harm its business and financial performance.
The Separation and Distribution Agreement provided that certain contracts, permits and other assets and rights were to be transferred from 3M or its subsidiaries to Solventum or its subsidiaries in connection with the separation. The transfer of certain of these contracts, permits and other assets and rights may have required consents or approvals of third parties or governmental authorities or provided other rights to third parties. In addition, in some circumstances, Solventum and 3M are joint beneficiaries of contracts, and Solventum and 3M may have needed the consents of third parties in order to split or separate the existing contracts or the relevant portion of the existing contracts to Solventum or 3M.
Some parties may use consent requirements or other rights to seek to terminate contracts or obtain more favorable contractual terms from Solventum, which, for example, could take the form of price increases. This could require Solventum to expend additional resources in order to obtain the services or assets previously provided under the contract or require Solventum to seek arrangements with new third parties or obtain letters of credit or other forms of credit support. If Solventum is unable to obtain required consents or approvals, it may be unable to obtain the benefits, permits, assets and contractual commitments that are intended to be allocated to Solventum as part of the Spin-Off, and Solventum may be required to seek alternative arrangements to obtain services and assets that may be more costly and/or of lower quality. The termination or modification of these contracts or permits or the failure to timely complete the transfer or separation of these contracts or permits could negatively affect Solventum’s business, financial condition, results of operations and cash flows.
Following the Spin-Off, Solventum’s commercial relationships with 3M remain significant, which could adversely affect Solventum’s business, its ability to meet other obligations and the market price of its common stock.
Following the Spin-Off, Solventum continues to have significant commercial relationships with 3M, including under the agreements that Solventum entered into with 3M in connection with the Spin-Off, which include the Separation and Distribution Agreement, a Transition Services Agreement, a Transition Distribution Services Agreement, a Transition Contract Manufacturing Agreement, Research and Development Master Services Agreements, Real Estate License Agreements, an Intellectual Property Cross License Agreement, a 3M Mark Use Agreement, a Transition Trademark License Agreement, Master Supply Agreements, a Tax Matters Agreement, an Employee Matters Agreement, and a Stockholder’s and Registration Rights Agreement (collectively, the "3M Agreements"), as well as 3M’s continued ownership of Solventum’s common stock and the ownership of equity interests of 3M by certain officers and directors of Solventum. These commercial relationships could potentially have important consequences to Solventum and its investors, including:
•Solventum could be negatively affected if it is required to make material payments pursuant to its indemnification obligations to 3M such as with respect to certain taxes (and any related costs and other damages) resulting from the separation and/or for uninsured liabilities related to the Bair Hugger patient warming system under the Separation and Distribution Agreement and certain other 3M Agreements. In addition, 3M’s indemnity of Solventum with respect to certain liabilities relating to PFAS prior to Spin-Off may not be sufficient to protect Solventum against the full amount of such liabilities if, for example, 3M fails to fully satisfy its indemnification obligations or disputes whether that liability is subject to indemnification.
•If Solventum does not satisfactorily perform its obligations under the 3M Agreements, it may be held liable for any resulting losses suffered by 3M, subject to certain limits. In addition, during the transition support periods under the Transition Services Agreement, the Transition Distribution Services Agreement and the Transition Contract Manufacturing Agreement, Solventum’s management and employees may be required to divert their attention away from its business in order to provide services to 3M.
•The terms of the 3M Agreements may be less beneficial to Solventum than the terms that would have resulted from arm’s-length negotiations between unaffiliated third parties, because the 3M Agreements were prepared in the context of the Spin-Off while Solventum was still a wholly owned subsidiary of 3M and did not have an independent Board of Directors or a management team that was independent of 3M.
•Solventum may incur temporary interruptions in business operations if it cannot transition effectively from 3M’s existing operating systems, databases and programming languages that support certain of its systems, including, for example, research and development support, information technology infrastructure and systems and accounting and reporting systems. The process of implementing an information technology infrastructure, in particular, has been and is expected to be expensive and time-consuming, and any difficulty, delay or additional expense incurred in developing such an infrastructure or transitioning from 3M’s information technology environment and systems could be disruptive to Solventum’s business operations and create risks to Solventum’s relationships with customers and other third parties. The failure to implement the new systems and transition data successfully and cost-effectively could disrupt Solventum’s business operations and have a material adverse effect on its profitability. In addition, these systems and
21
services may also be more expensive than the amounts reflected in its historical consolidated financial statements or less efficient than the systems and services 3M is expected to provide during the transition period to Solventum.
•Solventum is relying on 3M to satisfy its obligations under the 3M Agreements not only for a successful transition but also for the success of its long-term operations. If 3M does not, or is unable to, satisfy its obligations under some of the 3M Agreements, such as the Research and Development Master Services Agreements and the Master Supply Agreements, under which 3M has agreed to provide services or goods to Solventum for either a long-term duration or for a shorter transition period, as applicable, Solventum could experience significant short-term and long-term disruptions to its business or other operational difficulties or losses.
•The disposition by 3M of its remaining ownership interest in Solventum, which currently represents approximately 14.8% of the outstanding Solventum common stock, may be subject to various conditions, including receipt of any necessary regulatory and other approvals and the existence of satisfactory market conditions. These conditions may not be satisfied or 3M may decide for any other reason not to consummate the disposition and instead retain a significant ownership interest in Solventum for a period of time, not exceeding five years. Satisfying the conditions relating to such disposition may require actions that 3M has not anticipated. Any delay by 3M in completing the disposition could have a material adverse effect on the market price for Solventum common stock.
•Certain of Solventum’s directors and employees may have actual or potential conflicts of interest because of their financial interests in 3M or because of their previous positions with 3M, which could have implications for both Solventum and 3M. For example, conflicts of interest could arise in connection with the resolution of any dispute between Solventum and 3M regarding the terms of the 3M Agreements or out of any commercial arrangements that Solventum or 3M may enter into in the future.
•The 3M Agreements may limit Solventum’s ability to fully exploit certain intellectual property due to certain field of use restrictions on the licenses granted between the parties, which may permit 3M to compete with Solventum using intellectual property owned by or licensed to Solventum. Solventum’s ability to compete may be further impacted by the non-competition provisions contained in the Separation and Distribution Agreement, which prohibit Solventum from manufacturing and/or selling certain products, or from selling its products to certain end users for a specified period of time following the Spin-Off.
Any of the foregoing may adversely impact demand for Solventum’s products or cause Solventum to lose market share, and could result in operational disruptions or additional expense, all of which may have an adverse effect on Solventum’s business, financial condition, results of operations and cash flows or may adversely impact the market price of Solventum’s common stock.
Risks Related to Solventum’s Business
General Economic and Business Risks
Solventum’s results may be impacted by the effects of, and changes in, worldwide economic, political, regulatory, international trade and geopolitical conditions, war and other events beyond its control.
Solventum develops, manufactures, distributes and sells its products globally, and, accordingly, Solventum’s operations and the execution of its business strategies and plans are subject to global competition and economic and geopolitical risks that are beyond its control, such as, among other things, disruptions in financial markets, economic downturns, military conflicts, political changes and trends such as protectionism, economic nationalism or regionalism resulting in government actions impacting international trade agreements, imposing trade restrictions such as tariffs, and retaliatory countermeasures, changes in regulatory regimes that could restrict Solventum’s ability to manufacture and sell its products (including healthcare regulatory regimes), diminished or insufficient protection of intellectual property and government deficit reduction and other austerity measures in locations or industries in which Solventum operates. For example, changes in the policies or practices of government programs, authorities or agencies (e.g., Medicare and Medicaid in the U.S.) could adversely impact the amount of funding Solventum’s customers have for its products and services. Trade and tariff actions have had an impact on Solventum and its suppliers and may have material adverse effects on Solventum's business, financial condition, results of operations and cash flows. Further escalation of specific trade tensions, including those between the U.S. and China, or more broadly of global trade conflict, could adversely impact Solventum’s business and operations around the world. Solventum’s business is also impacted by social, political and labor conditions in locations in which Solventum or its suppliers or customers operate; adverse changes in the availability and cost of capital; monetary policy; interest rates; inflation; recession; commodity prices; currency volatility or exchange control; ability to expatriate earnings; and other laws and regulations in the jurisdictions in which Solventum or its suppliers or customers operate.
22
The global economy has been impacted by the conflicts between Russia and Ukraine and in the Middle East and other geopolitical events. The U.S. and other governments have imposed export controls on certain products and financial and economic sanctions on certain industry sectors and parties in Russia. 3M suspended operations of its subsidiaries, including those of Solventum’s business, in Russia in March 2022 and, in September 2022, committed to a plan to exit the related net assets in Russia, including those of Solventum’s business, through a sale of 3M’s Russian subsidiaries that was consummated in June 2023. Solventum has operations that source certain raw materials from suppliers in Russia and has experienced related supply disruption due to the conflict. These geopolitical tensions could result in, among other things, cyberattacks, further supply chain disruptions impacting downstream customers, higher energy costs, lower consumer demand and changes to foreign exchange rates and financial markets, any of which may adversely affect Solventum’s business and supply chain.
The deployment and use of artificial intelligence ("AI"), machine learning, or other emerging technologies in Solventum’s products and services, including as part of its research and development efforts, or its failure to adapt its products or services to industry trends and developments related to such technologies in a timely manner, or at all, could adversely affect Solventum’s business, financial condition, results of operations and cash flows.
Solventum has begun to deploy AI in its products and services, including as part of its research and development efforts, and Solventum expects to continue to explore additional uses as AI continues to develop. In addition, Solventum intends to devote significant resources to develop and deploy cloud, edge and software solutions in its healthcare solutions. For example, Solventum has incorporated AI into certain products in the Health Information Systems business segment and certain of its digital offerings and expects to continue to build AI into additional products. Any disruption or failure in the AI, machine learning, or other emerging technologies Solventum deploys or uses in its products and services, including as part of its research and development efforts, could adversely impact Solventum’s business, including as a result of flawed AI algorithms, insufficient or biased datasets, malfunctions or manipulations, unintentional release of confidential information, or product or service delays or recalls. In addition, any failure to successfully deploy or use such technologies in Solventum’s products and services, or to adapt to medical technology industry trends and developments related to such technologies in a timely manner (or at all), particularly as competitors incorporate such technologies into new and existing products and services, could adversely affect customer demand for Solventum’s products and its competitiveness. The deployment of AI, machine learning or other emerging technologies into Solventum’s products and services will require additional investment and increase its costs. Furthermore, the rapid advancement of technology, including AI, creates additional risks as Solventum includes such technology into its products and services, including from confidentiality, privacy, data protection, cybersecurity and compliance perspectives, and raises intellectual property issues and operational, technological and other concerns. Any of the above factors could adversely affect Solventum’s business, financial condition, results of operations and cash flows.
Public health crises may increase Solventum’s cost of doing business and disrupt Solventum’s operations.
Due to Solventum’s global operations, Solventum’s business is and will be impacted by public health crises, epidemics and pandemics in the locations in which Solventum or its suppliers or customers operate, and these events have adversely affected, and could in the future adversely affect, Solventum’s operations and financial performance. As a result of such events, we have in the past experienced, and in the future may experience, increased economic and demand uncertainty and could experience adverse impacts to Solventum’s operations, including its supply chain; its manufacturing and distribution capabilities; site shutdowns, workplace disruptions; restrictions on movement of people, raw materials and goods (both at our own facilities and at those of our customers and suppliers); global supply chain disruptions and price inflation. Solventum is not able to predict the impact of public health crises, epidemics or pandemics, which may have a material adverse effect on its business, cash flows, financial condition and results of operations.
Our brands are critical to our success, and damage to our reputation or our brands could adversely affect our business, results of operations or financial condition.
Our ability to compete successfully depends on the strength of our brands. Following the Spin-Off, Solventum now operates under its own brand and accordingly may no longer benefit from 3M’s long operating history, reputation and well-known brand. Developing and maintaining the reputation of our brands is a critical component of our ability to attract and retain employees and maintain and develop our relationships with consumers, customers, manufacturers, suppliers, distributors and other third-party partners, including healthcare professionals, influencers and other individuals with whom we have relationships. We believe employees, consumers, customers and third-party partners value the reputation and status of our brands. However, these efforts may not be successful, and the loss of benefits conferred by 3M’s brand recognition and reputation or the failure to maintain the value of our brands could impact our relationships or brand loyalty with employees, consumers, customers and third-party partners and otherwise adversely affect our business, results of operations or financial condition.
Our reputation and our brands could in the future be damaged by negative publicity, whether or not valid. Negative publicity could relate to our company, our brands, our products, our supply chain, our ingredients, our packaging, our sustainability and social impact practices, our employees or any other aspect of our business. Our reputation or our brands could
23
also be adversely affected by negative publicity related to our industry, our competitors, our competitors’ products, our customers or our third-party partners, including healthcare professionals, and other individuals with whom we have relationships, even if the publicity is not directly related to our company or our brands and even if the publicity is not accurate. In addition, widespread use of digital and social media platforms around the world has greatly increased the accessibility of information and the speed with which it is disseminated, which has made, and likely will continue to make, maintaining our reputation and our brands more challenging. Damage to our reputation or our brands could cause employees, consumers, customers and third-party partners to lose trust in our business or our products, require us to expend substantial resources to remedy the damage or otherwise adversely affect our business, results of operations or financial condition.
Acquisitions, strategic alliances, divestitures and other strategic events resulting from portfolio management actions and other evolving business strategies, and possible further organizational restructuring, could affect future results.
Solventum monitors its business portfolio and organizational structure and may make acquisitions, divestitures and changes to its organizational structure or enter into strategic alliances, equity investments or joint ventures. For example, on September 2, 2025, Solventum announced the close of its sale of the Purification & Filtration business ("P&F") to Thermo Fisher Scientific Inc. for $4.0 billion in cash before customary adjustments. In addition, on December 23, 2025, Solventum acquired Acera Surgical ("Acera"), a privately held bioscience company focused on developing and commercializing fully engineered materials for regenerative wound care, for cash consideration of $696 million, net of cash acquired, plus a future payment of $125 million dependent on the acquired business achieving a sales-based milestone. These activities have resulted, and any future activities may result, in substantial investment of Solventum’s resources. The success of any such activities will depend upon a number of factors, including Solventum’s ability to:
•identify suitable acquisition targets or assets, conduct due diligence, negotiate transactions on favorable terms and ultimately complete such transactions;
•compete for acquisition targets and assets, which may lead to substantial increases in purchase price or terms that are less attractive to Solventum;
•finance any future acquisition, investment, alliance or other transaction on terms acceptable to Solventum, if at all (which may involve the use of Solventum’s shares for payment of the purchase price);
•identify, negotiate and ultimately complete suitable divestitures or other strategic transactions;
•comply with applicable laws and regulations, including foreign laws and regulations;
•obtain any legally required rulings by antitrust or other regulatory bodies;
•successfully and timely integrate and operate acquired businesses;
•successfully separate any divested business;
•protect intellectual property and prevail in litigation relating to newly acquired technologies;
•predict or realize expected growth opportunities, cost savings, synergies and market acceptance of acquired companies’ products; and
•successfully identify and retain key target employees and customers.
In addition, acquisitions may expose Solventum to significant risks and uncertainties, including failure to identify significant non-compliant behaviors or practices by, or liabilities relating to, the acquisition target (or its agents) prior to acquisition; successor liability imposed by regulators for actions by the acquisition target (or its agents) prior to acquisition; and diversion of management’s attention from existing operations to the acquisition and integration process. Equity and other investments and strategic alliances pose additional risks, as Solventum could share ownership in companies and, in some cases, management responsibilities with one or more other parties whose objectives for the alliance may diverge from those of Solventum over time; who may not have the same priorities, strategies, or resources as Solventum does; or whose interpretation of applicable policies may differ from those of Solventum. Such transactions will be subject, in certain circumstances, to the consent of 3M under the Tax Matters Agreement that Solventum entered into with 3M, as discussed in "—Risks Related to the Spin-Off and Solventum’s Relationship with 3M." There can be no assurance that any future transactions of this type will be pursued or, if pursued, will be successful.
Solventum may not be able to effectively integrate acquired businesses into its operations or achieve expected cost savings or profitability from its acquisitions.
Solventum’s acquisitions involve numerous risks, including:
•unforeseen difficulties in integrating personnel and sales forces, operations, manufacturing, logistics, research and development, information technology, compliance, vendor management, communications, purchasing, accounting, marketing, administration and other systems and processes;
•difficulties harmonizing and optimizing quality systems and operations;
•diversion of financial and management resources from existing operations;
•unforeseen difficulties related to entering markets for which or geographic regions where Solventum does not have prior experience;
24
•potential loss of key employees;
•unforeseen risks and liabilities associated with businesses acquired, including any unknown vulnerabilities in acquired technology, compromises of acquired data or noncompliance with data privacy requirements; and/or
•inability to generate sufficient revenue or realize sufficient cost savings to offset acquisition or investment costs.
As a result, if Solventum fails to evaluate and execute acquisitions properly, Solventum might not achieve the anticipated benefits of such acquisitions, and Solventum may incur costs in excess of what it anticipates. These risks would likely be greater in the case of larger acquisitions.
Solventum’s restructuring program may not be successful or Solventum may not fully realize the expected cost savings and/or operating efficiencies from its restructuring initiatives.
Solventum has initiated a multiyear restructuring program, 'Transform for the Future', which is designed to further enable its long-term growth strategy and ensure it's best positioned to compete-to-win in a rapidly changing healthcare environment. Designed to transform the cost structure, enhance operational efficiency, and reposition for profitable growth, the primary activities of the restructuring program include operating structure optimization and workforce reorganization, procurement and cost management, supply chain, manufacturing and global footprint optimization, and streamlining systems and increased automation to improve operational efficiency. Additionally, as a result of the restructuring initiative, Solventum may experience a loss of continuity, loss of accumulated knowledge and/or inefficiencies during transitional periods. The restructuring initiative presents risks that may impair Solventum’s ability to achieve anticipated operating enhancements and/or cost reductions, or otherwise harm its business, including higher than anticipated costs in implementing the restructuring program, as well as management distraction. For more information on Solventum’s restructuring program, see Note 14 to Solventum’s consolidated financial statements. If Solventum fails to achieve some or all of the expected benefits of its restructuring program, or if costs related to the restructuring program are more than anticipated, it could have a material adverse effect on Solventum’s competitive position, business, financial condition, results of operations and cash flows.
Solventum may not be able to access the capital and credit markets on terms that are favorable to Solventum, or at all.
Solventum could need access to the capital markets to supplement its existing funds and cash generated from operations to satisfy its needs for working capital, to meet capital expenditure and debt service requirements, and for other business initiatives. Solventum’s ability to issue additional debt or enter into other financing arrangements on acceptable terms could be adversely affected by its debt levels, unfavorable changes in economic conditions or uncertainties that affect the capital markets. In the event of adverse capital and credit market conditions, Solventum may be unable to obtain capital market financing on favorable terms, or at all.
Change in Solventum’s credit ratings could increase cost of funding.
Solventum’s credit ratings are important to its cost of capital. The major rating agencies will routinely evaluate Solventum’s credit profile and assign debt ratings to Solventum. This evaluation is based on a number of factors, which include financial strength, business and financial risk, as well as transparency with rating agencies and timeliness of financial reporting. Changes in Solventum’s credit ratings could adversely affect its ability to obtain capital market financing and the cost of such financing. Moreover, a reduction in Solventum’s credit rating to below investment-grade could cause certain customers to reduce or cease to do business with Solventum, which would adversely impact its financial performance.
Foreign currency exchange rates and fluctuations in those rates may affect Solventum’s ability to realize projected growth rates in its sales and earnings.
Because Solventum’s financial statements are denominated in U.S. dollars and a material percentage of its revenues are derived from outside the U.S., Solventum’s results of operations and its ability to realize projected growth rates in sales and earnings could be adversely affected by volatility in foreign currency exchange rates, particularly if the value of the U.S. dollar strengthens significantly against foreign currencies. Following the Spin-Off, Solventum may be more exposed to matters such as foreign currency exchange rates as a smaller, standalone company than it had been as a part of the larger 3M enterprise.
Solventum cannot predict with any certainty changes in foreign currency exchange rates or its ability to mitigate these risks. Solventum may experience additional volatility because of increasing inflationary pressures and other macroeconomic factors, including in emerging market countries. Solventum may be unable to hedge the effects of foreign exchange rate changes in a cost-effective manner.
Changes in interest rates could adversely affect Solventum.
Solventum is exposed to changes in interest rates, including through variable rate debt and due to the fact that increases in interest rates may adversely affect the financial condition of Solventum’s counterparties in a manner that may affect their ability
25
to transact with Solventum or their demand for Solventum’s products and services. Any of the foregoing could adversely affect Solventum’s business, results of operations, financial condition and cash flows.
Market Dynamic Risks
Solventum operates in highly competitive markets, competition may increase in the future and the healthcare industry may be disrupted, necessitating that Solventum lower prices or resulting in a loss of market share.
Healthcare markets are characterized by rapidly evolving technology, frequent introduction of new products, intense competition and pricing pressure. Solventum faces substantial competition from international and domestic companies of all sizes, including existing competitors, new market entrants and non-traditional entrants. Demand for Solventum’s products and services, which impacts revenue and profit margins, will be affected by, among other things, (i) the development and timing of the introduction of competitive products and services; (ii) Solventum’s pricing strategies; (iii) changes in customer order patterns, such as changes in the levels of inventory maintained by customers, vendors or channel partners; (iv) changes in customers’ preferences for Solventum’s products and services, including the success of products and services offered by competitors; (v) changes in customer designs for their products and services that can affect the demand for Solventum’s products and services; (vi) the emergence of new AI-enabled competitors and business model disruption, particularly for our HIS business; (vii) changes in the business environment related to disruptive technologies, such as AI, block-chain, expanded analytics and other enhanced learnings from increasing volume of available data; (viii) local market conditions, such as mandatory intellectual property transfers, protectionist measures and other government policies supporting increased local competition; (ix) costs of production or delivery, whether due to geographic location, currency fluctuations, taxes, duties or otherwise; (x) the perception of Solventum’s brand and image in the market; (xi) changing regulatory standards, legal requirements or enforcement rigor; (xii) failure to acquire or effectively integrate businesses and technologies that complement or expand Solventum’s existing businesses; and (xiii) consolidation among customers, suppliers, channel partners or competitors.
In addition, Solventum’s inability to obtain and maintain regulatory authorizations for, and supply commercial quantities of, Solventum’s products and services as quickly and effectively as its competitors could limit market acceptance. Solventum’s competitors may also have greater financial, marketing and other resources, respond more quickly to new or emerging technologies, undertake more extensive marketing campaigns; adopt more aggressive pricing policies, or be more successful in attracting potential customers, employees and strategic partners.
Consolidation in the healthcare industry could have an adverse effect on Solventum’s revenues and results of operations.
Many healthcare industry companies, including healthcare systems, distributors, manufacturers, providers and insurers, are consolidating or have formed strategic alliances. As the healthcare industry consolidates, competition to provide products and services is expected to continue to intensify, resulting in pricing pressures, decreased average selling prices and the exclusion of certain suppliers from important market segments. Consolidations also create larger enterprises with greater negotiating power and Solventum may lose customers. If consolidation trends continue, it could adversely affect Solventum’s business results, cash flows, financial condition or prospects. If Solventum faces an increase in costs or reduces its prices because of industry consolidation, or if Solventum loses customers as a result of consolidation, its business, results of operations, financial condition and cash flows could be adversely affected.
Reductions in customers’ research budgets or government funding may adversely affect Solventum’s business.
Solventum’s customers include hospitals, universities, healthcare providers, government agencies and public and private research institutions. Research and development spending of such customers can fluctuate based on spending priorities and general economic conditions. The level of government funding of research and development is unpredictable. The availability of governmental research funding may be adversely affected by many factors, including public spending priorities, available resources, economic conditions and governmental spending reductions, particularly during periods of economic uncertainty. Stalemates in national, regional or local government budgeting decisions could also lead to substantial delays or reductions in governmental spending. Any reduction or delay in governmental funding could cause Solventum’s customers to delay or forgo purchases of its products.
Solventum’s growth objectives are largely dependent on the timing and market acceptance of its new products and services, including its ability to continually renew its pipeline of new products and services and to bring those products and services to market.
A significant element of Solventum’s strategy is to increase revenue growth by focusing on innovation and new product and service development. New service and product development requires significant investment in research and development, clinical trials and regulatory approvals. The ability to bring new products and services to market is subject to difficulties or delays in development, such as the inability to identify viable new products and services, obtain adequate intellectual property
26
protection, regulatory approvals and reimbursement in the U.S. and abroad and successfully complete clinical trials or gain market acceptance of new products and services. It is uncertain when or whether Solventum’s products or services currently under development will be launched or will be commercially successful. Additionally, new offerings may be quickly rendered obsolete by changing customer preferences, changing industry standards or competitors’ innovations or reverse engineering efforts. If Solventum cannot successfully introduce new products and services that address the needs of its customers, Solventum’s offerings may become obsolete, and its business results, cash flows and financial condition could suffer.
The success of many of Solventum’s products depends upon certain key healthcare professionals.
Solventum works with leading global healthcare professionals who provide considerable knowledge and experience. The research, development, marketing and sales of many of Solventum’s products depend on maintaining working relationships with healthcare professionals. Solventum relies on these professionals for assistance in the development and marketing of its products. A public health crisis, epidemic or pandemic, including any resurgence of the COVID-19 pandemic may limit access to these professionals, and resulting travel restrictions, shutdowns and similar measures in response to any such public health crisis may impact Solventum’s ability to maintain these relationships, which in turn would adversely affect its ability to develop, market and sell new and improved products. If new laws, regulations or other developments limit Solventum’s ability to appropriately engage these professionals or to continue to receive their advice and input or Solventum is otherwise unsuccessful in maintaining strong working relationships with these healthcare professionals, the success of Solventum’s products could suffer, which could result in a material adverse effect on its business, results of operations, financial condition and cash flows.
Changes in reimbursement practices of third-party payers or other cost containment measures or worsening economic conditions could affect the demand for Solventum’s products and the prices at which they are sold.
Sales of many of Solventum’s products directly or indirectly depend on the availability of reimbursement and the amount of reimbursement that its customers may seek from various third-party payers, including government programs, authorities or agencies (e.g., Medicare and Medicaid in the U.S.), and private health plans. The coverage policies and reimbursement levels of third-party payers, which can vary among public and private sources and by country, may affect which products customers purchase and the prices they are willing to pay for those products in a particular jurisdiction. Reimbursement rates can also affect the market acceptance rate of new technologies and products.
In general, employers and third-party payers, particularly in the U.S., have become increasingly cost-conscious, with higher deductibles imposed in many medical plans. Additionally, austerity measures or other reforms by foreign governments may limit, reduce or eliminate payments for Solventum’s products and adversely affect both pricing flexibility and demand for Solventum’s products. Even if Solventum develops promising new products, it may find limited demand for the products unless reimbursement approval is obtained from third-party payers. Further legislative or administrative reforms that impact reimbursements or pricing could result in a material adverse effect on its business, results of operations, financial condition and cash flows.
In addition, current or worsening economic conditions, including recessionary pressures, may adversely affect the ability of Solventum’s customers to pay for its products and services, and the amount spent on healthcare generally, which could result in decreased demand for Solventum’s products and services, declining cash flows, longer sales cycles, slower adoption of new technologies and increased price competition.
Pricing pressure has also increased due to continued consolidation among healthcare providers, trends towards managed care, the shift towards governments becoming the primary payers of healthcare expenses, reduction in reimbursement levels and medical procedure volumes and government laws and regulations relating to sales and promotion, reimbursement and pricing generally. As a result of these and other measures, including future measures or reforms that cannot be predicted, reimbursement may not be available or sufficient to allow Solventum to sell its products on a competitive basis. Legislation and regulations affecting reimbursement for Solventum’s products may change at any time. Solventum cannot predict the impact of these pressures and initiatives or any negative effects of any additional regulations that may affect its business.
Solventum’s future results are subject to vulnerability with respect to materials and fluctuations in the costs and availability of purchased components, compounds, raw materials, energy, production capacity and labor due to shortages, increased demand and wages, logistics, supply chain interruptions, manufacturing site disruptions, regulatory developments, tariffs, inflation and other disruptive factors.
Solventum depends on various components, compounds, raw materials and energy (including oil and natural gas and their derivatives) supplied by others for the manufacturing of its products. If suppliers fail to meet their delivery obligations, raise prices or cease to supply to Solventum, it may continue to cause delays in delivery or significantly increase Solventum’s costs. If Solventum loses suppliers, if their operations are substantially interrupted, if their prices continue to increase significantly due to tariffs or inflationary pressures, or if any of them fail to meet performance or quality specifications, Solventum may be
27
required to identify and qualify one or more replacement suppliers. This also may require Solventum to redesign or modify its products to incorporate new components and obtain regulatory authorization, qualification or certification of these redesigned or modified products.
Supplier relationships could be interrupted due to supplier material shortages, equipment malfunctions, transportation delays, tariffs, inflationary pricing pressures, work stoppages, labor shortages and other disruptive events, or could be terminated. In addition, some of Solventum’s suppliers are limited- or sole-source suppliers, and Solventum’s ability to meet its obligations to customers depends on the performance, product quality and stability of such suppliers and Solventum’s ability to source alternatives in a cost effective manner. Solventum may experience a sustained interruption in its receipt of adequate supplies or the distribution of its products, or disruption to key manufacturing sites’ operations, including due to government actions relating to discharge or emission permits or other legal or regulatory requirements. In addition, there can be no assurance that Solventum’s processes to minimize volatility in component and material pricing will be successful or that future price fluctuations or shortages will be managed. Solventum could incur contractual penalties, experience a deterioration in customer relationships or suffer harm to its reputation if it is unable to fulfill its obligations to customers. Any of the foregoing could have a material adverse effect on Solventum’s ability to fulfill supply obligations to its customers or on its business, results of operations, financial condition and cash flows.
Solventum’s business also depends on having sufficient production capacity to meet the demand for Solventum’s products and to support Solventum’s future growth. Solventum is conducting capital expansion efforts to increase its manufacturing capacity in certain areas of its operations. These efforts may not be successful. If this is the case, it could adversely affect Solventum’s operations and limit Solventum’s ability to support future growth or significantly impact its financial results and condition.
In addition, many of Solventum’s products require sterilization prior to sale, and Solventum utilizes contract sterilizers to perform this service. To the extent Solventum’s contract sterilizers are unable to sterilize Solventum’s products, whether due to capacity, availability of materials for sterilization, regulatory or other constraints, including federal and state regulations on the use of ethylene oxide, Solventum may be unable to transition to alternative internal or external resources or methods in a timely or cost effective manner or at all, which could have a material impact on Solventum’s results of operations and financial condition.
3M is the sole source of supply for raw materials used in certain of our products and our business will be harmed if 3M does not satisfy our requirements.
3M is the sole source of supply for certain chemical materials and inputs used in our products (including transparent IV film dressings, biological indicators for sterilization assurance, medical securement tapes and dental composites and cements) that, as of the Spin-Off, accounted for approximately $3 billion of our revenue for fiscal year 2025, including a material with a manufacturing process proprietary to 3M that is used in our products accounting for approximately $2 billion of our revenue for fiscal year 2025. While 3M has agreed to supply these items to Solventum for a period of time, our business will be harmed if 3M does not satisfy our requirements during such period of time, and Solventum may subsequently need to either reach agreement with 3M for an extended supply arrangement, develop our own manufacturing capabilities for these materials or identify an appropriate substitution or product reformulation in order to continue manufacturing and selling the applicable products. There is no guarantee that 3M will agree to continue to supply these materials following the term of the supply agreement on commercially reasonable terms or at all. At this time, Solventum has not identified an appropriate substitute input to replace the materials supplied by 3M, and Solventum does not currently have the capability to manufacture such materials itself. If 3M’s obligation to supply us with this material ends before we can develop or secure an alternative source of supply, the related product sales, which may be material, will be at risk. Any alternatives we pursue to mitigate this risk might also result in higher costs to source or produce the relevant products. Solventum’s failure to ensure a continuing supply of these materials or find acceptable substitutes, or the costs incurred by Solventum in connection with securing such continuing supply or finding such substitutes, could have a material adverse effect on Solventum’s business, including potentially the loss of revenue from the relevant products.
Legal and Compliance Risks
Solventum is subject to risks related to international, federal, state and local treaties, laws and regulations that are subject to change at any time, as well as compliance risks related to legal or regulatory requirements, contract requirements, policies and practices or other matters that require or encourage Solventum or its suppliers, vendors or channel partners to conduct business in a certain way. The outcome of legal and regulatory proceedings related to compliance with the treaties, laws, regulations and requirements could have a material adverse effect on Solventum’s business, results of operations, financial condition and cash flows.
Solventum operates globally, including in some jurisdictions that pose potentially elevated risks of fraud or corruption or increased risk of internal control issues, and is subject to risks related to international, federal, state and local treaties, laws and
28
regulations, including those involving product liability; antitrust; intellectual property; environmental, health and safety; tax; the FCPA and other anti-bribery laws; international import and export requirements and trade sanctions compliance; regulations of the U.S. Food and Drug Administration and similar foreign agencies; privacy laws and information security policies and regulations; and U.S. federal healthcare program-related laws and regulations including the False Claims Act, anti-kickback laws and the Physician Payments Sunshine Act. Solventum is also subject to compliance risks related to legal or regulatory requirements, contract requirements, policies and practices or other matters that require or encourage Solventum and its suppliers, vendors or channel parties, to conduct business in a certain way, and Solventum’s activity as well as the activity of such suppliers, vendors or channel parties could adversely affect Solventum’s business. In particular, Solventum is subject to legal risks with respect to the below laws and regulations:
•Antitrust - Regulatory authorities may have authority in the event of alleged non-compliance with applicable law to impose fines and sanctions on Solventum or to require changes or impose conditions on the way Solventum conducts business. Under certain circumstances, violations of antitrust laws could result in suspension or debarment of Solventum’s ability to contract with certain parties or complete certain transactions. In addition, an increasing number of jurisdictions also provide private rights of action for competitors or consumers to seek damages asserting claims of anti-competitive conduct. An adverse outcome under any such investigation or audit could subject Solventum to fines or criminal or other penalties.
•FCPA and other anti-bribery laws - The failure to comply with the FCPA and similar anti-corruption and anti-bribery laws could result in significant civil fines and penalties or criminal sanctions against Solventum. Because of the predominance of government-sponsored healthcare systems around the world, many of Solventum’s customer relationships outside of the U.S. are with governmental entities, the employees of which may be considered government officials under such laws. Many anti-corruption laws also prohibit bribery of private sector individuals, and thus extend far beyond interactions with government officials. Solventum is subject to the FCPA’s accounting provisions, which require Solventum to keep accurate books and records and to maintain an adequate system of internal accounting controls sufficient to provide reasonable assurances of management’s control, authority and responsibility over Solventum’s assets. Global enforcement of anti-corruption laws has increased substantially in recent years, with more frequent voluntary self-disclosure by companies, aggressive investigations (including coordinated investigations across countries and governmental authorities) and enforcement proceedings by U.S. and non-U.S. governmental agencies, and assessment of significant civil and criminal fines, penalties and other sanctions against companies and individuals. From time to time, Solventum or its affiliates receive reports, internally and externally, via various reporting channels, about business and other activities that raise compliance or other legal or litigation issues. Solventum has been in the past, and in the future could be, required to investigate such reports and cooperate with U.S. and foreign regulatory authorities in such investigations, audit, monitor compliance or alter its practices as part of such investigations. While Solventum maintains and implements U.S. and international compliance programs, including policies and procedures, training and internal controls designed to reduce the risk of noncompliance, Solventum’s employees, suppliers, vendors, channel partners or agents may violate such policies and procedures and engage in practices that contravene relevant laws and regulations. Any alleged or actual violations of these anti-corruption laws may subject Solventum to government scrutiny, criminal or civil sanctions and other liabilities, including exclusion from government contracting, and could disrupt its business, adversely affect its reputation and result in a material adverse effect on its business, results of operations, financial condition and cash flows.
•Environmental Laws - Solventum is subject to environmental, health and safety laws and regulations concerning, among other things, the generation, handling, transportation and disposal of hazardous substances or wastes, the remediation of hazardous substances or materials at various sites and emissions or discharges into the land, air or water. If Solventum or its suppliers violate these environmental laws and regulations, facilities could be shut down, and violators could be fined or otherwise sanctioned. New laws and regulations, violations of these laws or regulations, stricter enforcement of existing requirements or the discovery of previously unknown contamination could require Solventum to incur costs or could become the basis for new or increased liabilities that could be material.
•Anti-Kickback and False Claims Laws - Solventum’s products are purchased by healthcare providers that typically bill various third-party payers, such as governmental healthcare programs (e.g., Medicare, Medicaid and comparable non-U.S. programs), private insurance plans and managed care plans, for the healthcare services provided to their patients. As a result, Solventum’s products are subject to regulation regarding quality and cost by the U.S. Department of Health and Human Services, including the Centers for Medicare & Medicaid Services ("CMS"), as well as comparable state and non-U.S. agencies responsible for reimbursement and regulation of healthcare goods and services, including laws and regulations related to kickbacks, false claims, self-referrals and healthcare fraud. Many states have similar laws that apply to reimbursement by state Medicaid and other funded programs as well as in some cases to all payers. As a manufacturer of products reimbursable by federal healthcare programs, Solventum is subject to the Physician Payments Sunshine Act, which requires it to annually report certain payments and other transfers of value it makes to U.S.-licensed physicians and certain advanced practice providers, and U.S. teaching hospitals. Any failure to comply
29
with these laws and regulations could subject Solventum or its officers and employees to criminal and civil financial penalties.
•Data Privacy, Emerging Technology and Cybersecurity Laws - Because Solventum is a business with a significant global footprint, compliance with evolving regulations and standards in data privacy, emerging technologies, and cybersecurity may result in increased costs, compliance challenges and the threat of increased regulatory enforcement activity. Solventum’s business relies on the secure electronic transmission, storage and hosting of sensitive information, including personal information, protected health information, financial information, intellectual property and other sensitive information related to our customers and workforce. Solventum is required to comply with increasingly complex and changing legal and regulatory requirements that govern the collection, use, storage, security, transfer, disclosure and other processing of personal data, including for the development and use of AI, in the U.S. and in other countries. Examples of those laws include, but are not limited to, the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (collectively, HIPAA), various U.S. state consumer privacy laws that have become effective recently, the EU General Data Protection Regulation (Regulation (EU) 2016/679 (GDPR) and the United Kingdom's version of the same, the Lei Gerai de Proteção de Dados Pessoias (Brazil LGPD), China’s Personal Information Protection Law (PIPL), PRC Cybersecurity Law and Personal Data Cross Border Transfer Rule, the European Union's Artificial Intelligence Act and various other country-specific requirements at the state and federal level around the world. In addition, privacy laws and regulations are becoming stricter and may potentially impose additional requirements on Solventum’s business, and certain jurisdictions have implemented data localization laws which can be costly and operationally difficult to satisfy. Similarly, the legal and regulatory landscape surrounding AI is rapidly evolving and uncertain and includes an increasingly large amount of complex, stringent, and inconsistent global regulations, including with regard to certification requirements in many of the countries in which Solventum's customers are located. Solventum cannot be sure how these laws and regulations will be interpreted, enforced, or applied to its operations, products and services. In addition to the risks associated with enforcement activities and potential contractual liabilities, Solventum’s ongoing efforts to comply with evolving laws and regulations may be costly and require ongoing modifications or limitations to its products, services, policies, procedures and systems. If Solventum or third parties are unable to effectively manage the use of AI or other emerging technologies by employees and third party providers, Solventum or third parties may fail to adequately safeguard confidential personal data, or if such information or data are wrongfully used by Solventum or third parties or disclosed to unauthorized persons or entities, such an event could result in a material adverse effect on its business, results of operations, financial condition and cash flows.
•Sustainability and Climate Change - There has been increased focus in recent years by federal, international, state and local regulatory and legislative bodies to expand mandatory sustainability reporting, diligence and disclosure requirements, including the European Union’s Corporate Sustainability Reporting Directive (CSRD), legislation in California requiring reporting of greenhouse gas emissions and climate risk and similar regulatory requirements in other jurisdictions. Additionally, there is an increase in regulatory mandates to combat and/or limit the effects of climate change through a variety of means, including regulating greenhouse gas emissions (and the establishment of enhanced internal processes or systems to track them), policies mandating or promoting the use of renewable or zero-carbon energy and sustainability initiatives, and additional taxes on fuel and energy. If legislation or regulations are enacted or promulgated in the U.S. or in any other jurisdiction in which Solventum does business that impose more stringent restrictions and requirements than its current legal or regulatory obligations, Solventum and companies in its supply chain may experience increased compliance burdens and costs to meet the regulatory obligations, which could cause disruption in the sourcing, manufacturing and distribution of its products and adversely affect its business, results of operations, financial condition and cash flows. Furthermore, new regulatory mandates related to extended producer responsibility laws, single-use plastic or chemical bans, or similar product-focused legislation may cause shift in market trends. These outcomes may in turn result in customers transitioning to competitive products, loss of market share, negative publicity, reputational damage, loss of customer confidence or other negative consequences (including a decline in stock price).
Solventum’s results of operations could be adversely impacted if the costs to comply with these evolving treaties, laws, regulations and requirements are greater than projected by Solventum. In addition, the outcome of legal and regulatory proceedings related to compliance with these treaties, laws, regulations and requirements are difficult to reliably predict, may differ from Solventum’s expectations and can result in, among other things, government scrutiny; criminal, civil or administrative sanctions, including fines; limitations on the extent to which Solventum can conduct business; employee and business partner terminations due to policy violations; reputational damage; private rights of action that result in litigation exposure, including expenses and costs incurred in connection with settlement or court proceedings, for Solventum; and other liabilities. In some instances, Solventum may make self-disclosures to relevant authorities that may pursue or decline to pursue enforcement proceedings against it. In addition, detecting, investigating and resolving actual or alleged violations of these treaties, laws, regulations or requirements is expensive and could consume significant time and attention of Solventum’s senior management. Although Solventum maintains general liability insurance to mitigate monetary exposure, the amount of liability
30
that may result from certain of these risks may not always be covered by, or could exceed, the applicable insurance coverage. Various factors or developments can lead Solventum to change current estimates of liabilities and related insurance receivables where applicable or make such estimates for matters previously not susceptible of reasonable estimates, such as a significant judicial ruling or judgment, a significant settlement, significant regulatory developments or changes in applicable law. Conducting internal investigations or responding to audits or investigations by government agencies could be costly and time-consuming. A future adverse ruling, settlement or unfavorable development could result in future charges that could result in a material adverse effect on its business, results of operations, financial condition and cash flows in any particular period. In addition, negative publicity related to the matters noted above or other matters involving Solventum may negatively impact Solventum’s reputation.
Solventum may face potential liabilities related to PFAS, which could adversely impact Solventum’s results.
3M agreed to assume, and indemnify and defend us against, certain liabilities relating to PFAS, generally including all such liabilities relating to the period prior to the Spin-Off, as well as certain liabilities relating to Solventum products that contain PFAS and that continue to be sold on the same basis by Solventum following the Spin-Off through 2025. As an independent company, Solventum is generally responsible for all PFAS-related liabilities resulting from its business, operations and activities following the Spin-Off, subject to the indemnification by 3M of certain liabilities relating to certain Solventum products sold through 2025. See "—Risks Related to the Spin-Off and Solventum’s Relationship with 3M—Following the Spin-Off, Solventum’s commercial relationships with 3M remain significant, which could adversely affect Solventum’s business, its ability to meet other obligations and the market price of its common stock."
Certain of Solventum’s products, like the products of other companies in Solventum’s industries, contain or are enabled by PFAS. 3M announced in December 2022 that it would work to discontinue the use of PFAS across its product portfolio by the end of 2025, and, as described above, 3M agreed to indemnify Solventum for certain PFAS-related product claims related to sales of products through such date. Solventum has also taken actions to meet this goal while owned by 3M, and, except as described below, Solventum intends to minimize the use of PFAS in products wherever feasible. 3M ceased supplying Solventum with PFAS-related products or components at the end of 2025.
Solventum continues to evaluate the availability of third-party components and products that do not contain PFAS in connection with working towards the goal of discontinuing the use of PFAS in its products. Depending on the availability and feasibility of such third-party components and products not containing PFAS (including the ability to obtain any required regulatory approvals), Solventum expects there will be some circumstances in which the use of PFAS-containing materials manufactured by third parties and used in certain applications in Solventum’s product portfolio. PFAS use is primarily limited to industry-standard fluoropolymers in third-party purchased components. Examples include o-rings, gaskets and seals used in filters needed for high stress process conditions, molded plastic parts used in the plastic housing of perioperative temperature management devices, medical grade membranes used in negative pressure wound therapy fluid canisters, release liners used with adhesive dressings, circuit boards used in electronic hardgoods, such as negative pressure wound therapy devices, and lithium ion batteries included in surgical clippers. Sales of these types of products will continue beyond 2025, in which case the 3M indemnification described above would not be available with respect to sales of products containing such materials after 2025. In such instances, Solventum intends to continue to evaluate the adoption of third-party products and components that do not contain PFAS to the extent such products and components become available and such adoption is feasible. Many companies in Solventum’s industries use PFAS-containing products and components, and Solventum believes that its use of such products and components is of a nature and magnitude that is broadly consistent with that of other companies in these industries.
There have been accelerating regulatory and legislative activities concerning PFAS globally, including increasingly strict restrictions on various uses of PFAS in products, as well as increased litigation relating to PFAS being filed against other parties. The potential options available to Solventum following 2025 (after which sales by Solventum of products containing or enabled by PFAS will no longer be subject to indemnification from 3M), regarding PFAS in products will involve risks, which risks may be material, and could have a material adverse effect on Solventum’s results of operations, cash flows or consolidated financial position.
Climate change and the impacts of climate change may materially adversely affect Solventum’s business, results of operations, financial condition and cash flows.
The impacts of climate change may include physical risks (e.g., rising sea levels or frequency and severity of extreme weather conditions, including natural disasters), social and human effects (e.g., population dislocations, economic disruption, political and social instability or harm to health and well-being) and transition risks (e.g., regulatory or technology changes), shifts in market trends (e.g., customers increasingly prioritize purchasing products that are sustainably made) and other adverse effects. In addition, Solventum faces the risk of increased compliance costs due to regulation aimed at eliminating carbon intensive inputs to its products; imposing climate-related costs associated with product sterilization, disposal, or recycling; and/or imposing an economic cost on carbon such as through carbon taxes or cap-and-trade systems.
31
Such impacts may cause physical damage to Solventum’s facilities as well as those of its suppliers, customers and other business partners, and disrupt Solventum’s supply chain and operations by adversely affecting its ability to procure goods or services required for the operation of its business at the quantities and levels it requires due to impairment of the availability and cost of certain products, materials, natural resources, commodities and energy.
Additionally, the impacts of climate change may further influence customer preferences and requirements, such as increased demand for products with lower environmental footprints, and for companies to produce and demonstrate progress against greenhouse gas reduction plans and targets. Failure to provide climate-friendly products or demonstrate greenhouse gas reductions could result in loss of market share.
Solventum operates in a strictly regulated industry, and compliance with laws and regulations applicable to the commercialization of Solventum’s products is costly and failure to comply may result in significant penalties.
The products Solventum develops, manufactures and commercializes are regulated in most of the markets Solventum serves. These regulations and standards govern the methods and controls used for the design, manufacture, packaging, labeling, storage, safety, sales and distribution, marketing clearance or approval, advertising and promotion, sterilization, installation, servicing, performance and effectiveness of the products Solventum sells globally. These regulations and standards apply to all facilities of Solventum’s business that conduct the activities previously described, regardless of where the facilities are located, as well as the activities performed by most of Solventum’s employees, including but not limited to, sales and marketing, research and development, regulatory affairs, quality assurance, medical, affairs and operations, both before and after a product is commercially distributed. Importantly, these regulations are promulgated and enforced by government bodies in individual countries, and differ by country and/or region and are dynamic.
Solventum commits a significant amount of resources to maintain compliance with these regulations and standards, which requires Solventum to create systems, processes and procedures that are aligned with the regulations in all markets Solventum serves. Compliance also requires Solventum to maintain knowledge of the current regulations that govern its activities. As these regulations change, Solventum must adapt its systems, processes and procedures to comply with the new regulations.
Governing bodies monitor compliance, among other ways, by conducting regularly occurring and unexpected audits of Solventum’s facilities to determine if Solventum’s systems, processes and procedures comply with the current regulations in the markets it serves. After each audit, the governing body typically provides a report of their findings describing the observations made during the audit. Sometimes these observations describe minor non-compliance issues in Solventum’s systems, processes and procedures. Often, these gaps require commensurate modifications but have no impact to Solventum’s ability to continue operations and commercialization of its products. In rare situations, the governing body may find significant or major non-compliance issues in Solventum’s systems, processes and procedures. If a governing body concludes, through these audits or otherwise, that Solventum is not in compliance with applicable laws or regulations or that any of its products are defective, ineffective or pose an unreasonable risk for patients, users or others, the governing body may require Solventum to recall a product or products, retract promotional materials and/or cease shipment of products, among other required actions; these requirements may remain in place until Solventum can demonstrate adequate compliance. Failure to demonstrate adequate modifications to Solventum’s systems, processes and procedures and continued compliance or repeat findings may result in more significant enforcement actions including but not limited to: warning letters, revocation of product approvals and licenses, injunctions, product seizure, penalties and fines, consent decrees and criminal prosecution, among other actions. These actions may have a negative impact on Solventum’s capital expenditures, earnings and competitive position.
To market its products internationally in compliance with applicable medical device and pharmaceutical regulations and standards, Solventum must obtain and maintain approvals for products and product modifications. The regulations promulgated by the governing bodies also require Solventum to submit data to demonstrate that its products meet the safety and effectiveness requirements to support the intended uses described in its labeling. This data is reviewed by the governing bodies to determine if Solventum has provided the necessary and sufficient information to demonstrate the safety and effectiveness of its products for the intended use described in its labeling. In many cases, the governing bodies request additional information to make this determination, which sometimes requires Solventum to conduct new testing or clinical trials, delaying the approval and commercialization of the product. In rare instances, the governing body may disapprove the application, prohibiting Solventum’s ability to commercialize the product in that market. In these instances, Solventum may decide to cease commercialization efforts for the product in that (or those) market(s) or it may decide to modify the product or retest the products and resubmit the data to the governing bodies. Delays to products approvals or disapproval of Solventum’s applications may have a negative impact to Solventum’s capital expenditures, earnings and competitive position. Solventum’s failure to properly maintain approvals for its products may result in its inability to continue to commercialize products in that market.
This global regulatory environment will likely continue to evolve, which could impact Solventum’s ability, or increase the time and cost, to obtain future approvals for its products. The process of obtaining regulatory clearances and/or approvals to
32
market and sell Solventum’s products can be rigorous, costly and time-consuming and the clearances and/or approvals might not be granted timely or result in limitations on the indicated uses of products.
Regulatory authorities, including the FDA, also strictly regulate the indications for use and associated promotional safety and effectiveness claims that may be made about approved or cleared products. If regulatory authorities determine that Solventum has promoted or marketed a product for off-label use, including through external-facing materials, oral statements or physician training, Solventum could be subject to penalties, fines, injunctions, criminal prosecution or other penalties.
Solventum is subject to laws and regulations governing government contracts, public procurement and government reimbursements in many jurisdictions, as to which the failure to comply could adversely affect Solventum’s business.
Solventum sells its products to government entities throughout the world and will be directly or indirectly subject to government policies governing reimbursement for healthcare procedures and services and various statutes and regulations in a variety of jurisdictions that apply to companies doing business with the government. The laws governing government contracts can differ from the laws governing private contracts and government contracts may contain terms and conditions that are not applicable to private contracts or that expose Solventum to higher levels of risk and potential liability than non-government contracts. Similarly, most jurisdictions have public procurement laws and reimbursement policies that set out rules and regulations for purchases and reimbursements by governmental entities. Solventum’s failure to comply with these laws could result in contract terminations, suspension or debarment from contracting with these entities, civil fines and damages, criminal prosecution and possible exclusion from participation in federal healthcare programs such as Medicare and Medicaid, as well as possible recoupment of any overpayments related to such violations. These jurisdictions may modify their laws, policies, rules or regulations, or impose new requirements that adversely affect Solventum’s business.
Additionally, some governmental entities, including the U.S. federal government, can terminate contracts for their convenience or for Solventum’s default. These governmental entities may also be subject to continued legislative funding approval. Early termination for convenience of one or more of Solventum’s contracts, or a change in a government customer’s funding levels, could impact Solventum’s expected revenues. Early termination for default of one or more of Solventum’s contracts could subject it to penalties and damages resulting from the default, including costs for the governmental entity to reprocure the items under contract, in addition to other penalties previously listed.
Solventum will also be subject to government audits, investigations and oversight proceedings. Efforts to ensure its business arrangements comply with applicable laws will involve substantial costs. If any actions by governmental or enforcement authorities are instituted against Solventum, defense can be costly, time-consuming and may require significant financial and personnel resources. If Solventum is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business, including the imposition of civil, criminal and administrative penalties, damages, disgorgement, monetary fines, individual imprisonment, possible exclusion from participation in certain government healthcare programs (including Medicare and Medicaid in the U.S.), contractual damages, reputational harm, diminished profits and future earnings, and curtailment or restructuring of its operations. In addition, any of Solventum’s government contracts could be terminated or Solventum could be suspended or debarred from all government contract work.
Solventum is exposed to risks associated with product liability claims, including existing claims and claims resulting from the actions or inactions of its customers or third parties, and product recalls or safety alerts that are outside of its control.
Solventum is exposed to potential product liability risks that are inherent in the design, manufacture and marketing of medical technologies. Customers or their patients have brought and likely will continue to bring product liability claims if Solventum’s products fail, or allegedly fail, to perform as expected or show a failure rate that is higher than expected, or the use of Solventum’s products results, or is alleged to result, in bodily injury, death or property damage. Even if these or similar claims are without merit, they can result in costly and time-consuming litigation. Solventum may also be exposed to claims or regulatory action if its products do not conform or are alleged not to conform to applicable product or design specifications, labeling or manufacturing requirements. Product and other liability actions, claims or injunctions are subject to significant uncertainty and may be expensive, time-consuming and disruptive to Solventum’s operations. For these and other reasons, Solventum may choose to settle product liability claims and other liability actions, regardless of their actual merit. If any such action or injunction were finally determined adversely to Solventum, such decision could result in significant damages and reputational harm, including the possibility of punitive damages, and Solventum’s financial position could be adversely affected. Adverse publicity could result in additional regulation of Solventum’s products or the healthcare industry in general, delay regulatory approval of new products, cause reputational harm and adversely affect Solventum’s ability to promote, manufacture and sell its products, even if the claims against Solventum are later shown to be unfounded or unsubstantiated.
In addition, manufacturing or design defects, component failures, unapproved or improper use of Solventum’s products, or inadequate disclosure of risks or other information relating to the use of Solventum’s products could lead to injury or other serious adverse events. Such events could lead to recalls or safety alerts relating to Solventum’s products (either voluntary or as required by the FDA or similar governmental authorities in other countries), and could result, in certain cases, in the removal of
33
a product from the market. A recall could result in significant costs and lost sales and customers, enforcement actions and/or investigations by state and federal governments or other enforcement bodies, as well as negative publicity and damage to Solventum’s reputation that could reduce future demand for its products.
Information Technology and Intellectual Property Risks
Solventum employs information technology systems to support its business and collect, store and use proprietary and confidential information. Security and data breaches, cyberattacks and other cybersecurity incidents involving Solventum’s information technology systems and infrastructure could disrupt or interfere with Solventum’s operations; result in the compromise and misappropriation of proprietary and confidential information belonging to Solventum or its customers, suppliers and employees; and expose Solventum to numerous expenses, liabilities and other negative consequences, including violations of applicable laws, any or all of which could adversely impact Solventum’s business, reputation and results of operations.
In the ordinary course of business, Solventum relies on centralized and local information technology networks and systems, some of which are provided, hosted or managed by vendors and other third parties, to process, transmit and store electronic information, and to manage or support a variety of businesses. That technology includes systems that could be used to process, transmit and store sensitive information, including personal information, protected health information, employee data, financial information, intellectual property, clinical data and sales and marketing data. Further, Solventum expects that the breadth and complexity of its information technology networks and systems will increase as it expands its product and service offerings to utilize AI, machine learning, and other emerging technologies. Third parties and threat actors, including organized criminals, nation-state or nation-state supported actors who are increasingly well-resourced, regularly attempt to gain unauthorized access to the information technology networks and infrastructure, data and other information used by or belonging to Solventum, and many such attempts are increasing in their frequency, sophistication and intensity and are not recognized until launched against a target. In addition, AI is increasing in use among such third parties and threat actors which may lead to more sophisticated and targeted phishing attempts, cyberattacks, phishing attempts, or otherwise enhance the social engineering capabilities of such parties. Despite Solventum’s cybersecurity and business continuity measures (including employee and third-party training, monitoring of networks and systems, patching, maintenance and backup of systems and data), its information technology networks and infrastructure are still potentially susceptible to attack, compromise, damage, disruption or shutdown, including as a result of the exploitation of known or unknown hardware or software vulnerabilities in its systems, the introduction of computer viruses or ransomware, service or cloud provider disruptions or security breaches, phishing attempts, employee error or malfeasance, power outages, telecommunication or utility failures, systems failures, natural disasters or other catastrophic events. Furthermore, Solventum relies on third-party vendors to supply and/or support certain aspects of its information technology systems and resulting products. These third-party systems could also become vulnerable to cyber-attack, malicious intrusions, breakdowns, interference or other significant disruptions, and may contain defects in design or manufacture or other problems that could result in system disruption or compromise the information security of Solventum’s own systems. Solventum’s increased adoption of remote working also introduces additional threats and risk of disruptions to its information technology networks and infrastructure. Geopolitical conflict may increase cybersecurity risks on a global basis.
As noted elsewhere in these Risk Factors, Solventum is using AI, machine learning, cloud, edge, and other emerging technologies in its products and services. Use of these technology solutions could result in Solventum’s increased vulnerability to cyber-attack, malicious intrusions, breakdowns, interference or other significant disruptions, exposure to penalties from non-compliance with emerging regulations, and may result in defects in design or manufacture or other problems that could result in system disruption or compromise the information security of Solventum’s other systems.
Despite Solventum’s cybersecurity measures, it is possible for security vulnerabilities or a cyberattack to remain undetected for an extended time period, up to and including several years, and the prioritization of decisions with respect to security measures and remediation of known vulnerabilities that Solventum and the vendors and other third parties upon which Solventum relies may prove inadequate to protect against attacks. While Solventum may experience future cyberattacks on and disruptions of its information technology systems and infrastructure, Solventum is not aware of any such incidents to date having had a material impact.
If Solventum’s information technology systems, products or services or sensitive data are compromised, there are many consequences that could result including, but not limited to, patients or employees being exposed to financial or medical identity theft or suffering a loss of product functionality; losing existing customers or having difficulty attracting new customers; experiencing difficulty preventing, detecting and controlling fraud; being exposed to the loss or misuse of confidential information; having disputes with customers, physicians and other healthcare professionals; experiencing increases in operating expenses or an impairment in its ability to conduct its operations; incurring expenses or losing revenues as a result of a data privacy breach, product failure, information technology outages or disruptions; voluntary or forced recalls or modifications to Solventum’s products; or suffering other adverse consequences including time-consuming and expensive lawsuits or other legal action and damage to Solventum’s reputation.
34
Solventum is subject to numerous international, federal and state privacy and security laws. Security and data breaches, cyberattacks and other cybersecurity incidents involving data protected by such laws, such as patient medical records and other health information, could subject Solventum to onerous governmental and regulatory investigations, fines and remediation actions, in addition to private litigation by affected individuals.
Solventum may be unable to obtain, maintain, protect or effectively enforce its intellectual property rights.
Solventum is substantially dependent on patent and other proprietary rights and relies on a combination of patents, trademarks, tradenames, copyrights, trade secrets and agreements (such as employee, non-disclosure and non-competition agreements) to protect its business and proprietary intellectual property. However, Solventum cannot assure that its means of obtaining, maintaining and enforcing its intellectual property rights will be adequate to maintain a competitive advantage.
Intellectual property laws differ in various jurisdictions in which Solventum operates and are subject to change at any time, which could further restrict Solventum’s ability to protect its intellectual property and proprietary rights. In particular, a portion of Solventum’s revenues is derived from jurisdictions where adequately protecting intellectual property rights may prove more challenging or impossible. In addition, the laws of many jurisdictions may not provide an adequate forum to effectively address situations where Solventum’s intellectual property rights have been compromised.
Protecting against the unauthorized use of proprietary technology is difficult and expensive and Solventum may need to litigate with third parties to enforce or defend patents issued to it or to determine the enforceability and validity of its proprietary rights or those of others. Determining whether an offering infringes, misappropriates or otherwise violates a third party’s intellectual property rights involves complex legal and factual issues, and the outcome of this type of litigation is often uncertain and inconsistent.
At any given time, Solventum may be involved as either plaintiff or defendant in a number of patent infringement actions, the outcomes of which may not be known for prolonged periods of time. While it is not possible to predict the outcome of patent and other intellectual property litigation, such litigation could result in Solventum’s payment of significant monetary damages and/or royalty payments, negatively impact Solventum’s ability to sell current or future products, or prohibit it from enforcing its patent and proprietary rights against others, which could result in a material adverse effect on its business, results of operations, financial condition and cash flows. Regardless of the merits or outcome, the resolution of any intellectual property dispute could require significant financial and management resources.
Solventum may not receive protection for pending or future applications relating to intellectual property rights owned by or licensed to it and the claims allowed under any issued intellectual property rights may not be sufficiently broad to protect Solventum’s products, services, solutions and any associated trademarks. Products sold by Solventum’s competitors may infringe, misappropriate or otherwise violate intellectual property rights owned or licensed by Solventum and such infringement, misappropriation or violation may be undetected by Solventum. In addition, as Solventum’s patents will expire in the normal course. Solventum’s inability to protect its intellectual property could result in a material adverse effect on its business, results of operations, financial condition and cash flows.
Additionally, Solventum licenses or will license certain intellectual property owned by 3M. As 3M is the owner of such intellectual property, Solventum will need to engage with 3M regarding the enforcement of such intellectual property rights. To the extent 3M and Solventum cannot align on prosecuting infringement of the intellectual property licensed to Solventum, such intellectual property may not be protected and continued infringement of such intellectual property could result in a material adverse effect on Solventum’s business, results of operations, financial condition and cash flows.
Tax Matters Risks
Changes in tax rates, laws or regulations could adversely impact Solventum’s financial results.
Solventum’s business is subject to tax-related external conditions, such as tax rates, laws and regulations in the U.S. and foreign jurisdictions. Changes in tax rates, laws or regulations, including further developments arising from tax reform legislation or regulation in the U.S. or foreign jurisdictions, could impact Solventum’s financial statements.
In particular, Solventum could be negatively impacted by the Base Erosion and Profit Shifting 2.0 initiative ("BEPS 2.0") by the Organization for Economic Cooperation and Development ("OECD") which, if enacted by OECD member countries, would likely impact the amount of tax that multinationals, such as Solventum, pay in the future.
Due to the uncertainty of any tax changes and other tax-related factors at this time, it is currently not possible to assess the ultimate impact these actions may have on Solventum’s financial statements. Solventum intends to monitor BEPS 2.0 and other
35
tax-related developments in the U.S. and foreign jurisdictions, including rule changes and implementation timing, to evaluate the impact BEPS 2.0 and other tax legislation or regulation may have on Solventum’s financial results.
Solventum’s tax burden could increase as a result of ongoing or future tax audits and inquiries.
Solventum is subject to periodic tax audits and inquiries by tax authorities. Tax authorities may disagree with Solventum’s interpretation of applicable tax laws and regulations, which could result in examination from taxing authorities and additional taxes. Solventum regularly assesses the likely outcomes of these tax audits in order to determine the appropriateness of Solventum’s tax provision. However, Solventum may not accurately predict the outcomes of these tax audits and, as a result, the ultimate outcome of any of these examinations could have a retroactive or prospective impact on Solventum’s overall tax burden.
Solventum could be negatively impacted by future changes in the allocation of income to each of the income tax jurisdictions in which Solventum operates.
Solventum operates in multiple income tax jurisdictions both in the U.S. and internationally. Solventum has adopted transfer pricing policies that determine how income is allocated to each of the income tax jurisdictions in which Solventum operates, based on current interpretations of complex income tax regulations. The allocation of Solventum’s income could be impacted by different factors including tax law changes, tax audits, underlying business changes, organizational changes, or operating model changes, which could result in increases to Solventum’s overall tax burden.
Solventum may benefit from various global tax incentives intended to encourage investment or employment; if Solventum’s incentives are not renewed or Solventum cannot or does not wish to satisfy all or part of the tax incentive conditions, Solventum may lose the tax incentives and could be required to refund tax incentives previously realized.
Solventum benefits or may benefit in the future from various global tax incentives intended to encourage investment or employment. If Solventum’s incentives are not renewed or Solventum cannot or does not wish to satisfy all or part of the tax incentive conditions, Solventum may lose the tax incentives and could be required to refund tax incentives previously realized or granted. As a result, Solventum’s tax burden could be higher than it would have been had Solventum maintained the benefits of the tax incentives.
Employee Matters Risks
If Solventum is unable to attract or retain key personnel and qualified employees, or maintain relations with its employees, unions and other employee representatives, Solventum’s business would be adversely affected.
There is substantial competition for key personnel, senior management, research and development personnel, and qualified employees in the healthcare industry and Solventum may face increased competition for such a highly qualified scientific, technical, clinical and management workforce in a highly competitive environment. Solventum’s ability to recruit and retain such talent will depend on a number of factors, including how its compensation, benefits, work location and work environment compare with those offered by its competitors and other local employers. There can be no assurance that Solventum will be successful in retaining existing personnel or recruiting new personnel.
The loss of one or more key employees, inability to attract or develop additional qualified employees, any delay in hiring key personnel, any deterioration of the relationships with its employees, or any material work stoppage, strike, or similar action could result in a material adverse effect on its business, results of operations, financial condition and cash flows.
Risks Related to Solventum Common Stock
A significant number of shares of Solventum common stock may be sold by 3M or others, which may cause the Solventum stock price to decline.
Any sales of substantial amounts of Solventum common stock in the public market or the perception that such sales might occur may cause the market price of Solventum common stock to decline. Shares distributed to 3M shareholders in the separation are generally freely tradeable without restriction or further registration under the U.S. Securities Act of 1933, as amended (the "Securities Act"), except for shares owned by Solventum’s "affiliates," as that term is defined in Rule 405 under the Securities Act. Accordingly, 3M shareholders may sell large amounts of Solventum common stock in the open market, and Solventum is unable to predict whether and when such sales would occur.
3M holds 14.8% of Solventum’s outstanding common stock. Based solely on statements as part of the Spin-Off, the Company believes 3M plans to dispose of all of the Solventum common stock that it retains after the Spin-Off through one or more sales of such shares (not later than five years after the Spin-Off). Solventum agreed that, upon the request of 3M and
36
pursuant to the terms of the stockholder’s and registration rights agreement, it will use its reasonable best efforts to effect a registration under applicable federal and state securities laws of any shares of Solventum’s common stock retained by 3M to the extent that 3M wishes to sell the shares of our common stock it retained in a registered offering. 3M’s shares are restricted securities within the meaning of Rule 144 under the Securities Act and are eligible for resale by 3M in the public market without registration subject to volume, manner of sale and holding period limitations under Rule 144 under the Securities Act. Any sales of substantial amounts of Solventum common stock in the public market by 3M or the perception that such sales might occur, in connection with the Spin-Off or otherwise, may cause the market price of Solventum common stock to decline.
Because Solventum does not currently intend to pay any dividends on its common stock, holders of its common stock must rely on stock appreciation for any return on their investment. Solventum cannot guarantee the timing, declaration, amount or payment of dividends on its common stock.
Solventum does not currently anticipate paying a regular dividend on its common stock in the near future. The timing, declaration, amount and payment of any future dividend will be within the discretion of Solventum’s Board of Directors and will depend upon many factors, including Solventum’s financial condition, earnings, capital requirements of its operating subsidiaries, covenants associated with certain of Solventum’s debt service obligations, legal requirements, regulatory constraints, industry practice, ability to access capital markets, and other factors deemed relevant by Solventum’s Board of Directors. Moreover, if Solventum determines to pay any dividend in the future, there can be no assurance that it will continue to pay such dividends or the amount of such dividends.
Anti-takeover provisions could enable Solventum’s Board of Directors to resist a takeover attempt by a third party and limit the power of its shareholders.
Solventum’s amended and restated certificate of incorporation and amended and restated bylaws contain, and Delaware law contains, provisions that are intended to deter coercive takeover practices and inadequate takeover bids by making such practices or bids unacceptably expensive to the bidder and to encourage prospective acquirers to negotiate with Solventum’s Board of Directors rather than to attempt a hostile takeover. These provisions include, among others:
•until the annual stockholder meeting in 2028, Solventum’s Board of Directors will be divided into classes, which could have the effect of making the replacement of incumbent directors more time consuming and difficult;
•as long as Solventum’s Board of Directors is classified, Solventum directors can be removed by stockholders only for cause;
•Solventum’s Board of Directors have the sole authority to fix the size of Solventum’s Board of Directors;
•Solventum’s Board of Directors have the authority to amend and repeal Solventum’s amended and restated bylaws without a stockholder vote;
•Solventum’s shareholders do not have a right to call a special meeting or act by written consent;
•Solventum’s amended and restated certificate of incorporation and amended and restated bylaws do not provide for cumulative voting in the election of directors;
•Solventum’s Board of Directors have the power to designate and issue, without any further vote or action by the Solventum shareholders, shares of preferred stock from time to time in one or more series;
•Solventum’s shareholders have to follow certain procedures and notice requirements in order to present certain proposals or nominate directors for election at shareholder meetings; and
•Solventum’s amended and restated certificate of incorporation contains exclusive forum provisions (as described in more detail in the following risk factor).
In addition, Solventum is subject to Section 203 of the Delaware General Corporation Law, which could have the effect of delaying or preventing a change of control that you may favor. Section 203 provides that, subject to limited exceptions, persons that acquire, or are affiliated with persons that acquire, more than 15% of the outstanding voting stock of a Delaware corporation may not engage in a business combination with that corporation, including by merger, consolidation or acquisitions of additional shares, for a three-year period following the date on which that person or any of its affiliates becomes the holder of more than 15% of the corporation’s outstanding voting stock.
Solventum believes these provisions will protect Solventum shareholders from coercive or otherwise unfair takeover tactics by requiring potential acquirers to negotiate with Solventum’s Board of Directors and by providing Solventum’s Board of Directors with more time to assess any acquisition proposal. These provisions are not intended to make Solventum immune from takeovers; however, these provisions will apply even if the offer may be considered beneficial by some shareholders and could delay or prevent an acquisition that Solventum’s Board of Directors determines is not in the best interests of Solventum and its shareholders. These provisions may also prevent or discourage attempts to remove and replace incumbent directors.
37
In addition, an acquisition or further issuance of Solventum stock could trigger the application of Section 355(e) of the Code, causing the Spin-Off to be taxable to 3M. Under the Tax Matters Agreement, Solventum would be required to indemnify 3M for the resulting tax, and this indemnity obligation might discourage, delay or prevent a change of control that Solventum shareholders may consider favorable.
Item 1B. Unresolved Staff Comments
None.
Item 1C. Cybersecurity
Risk Management
We identify and assess cybersecurity risk through various technologies, processes and policies that are regularly updated to align with the changing threat landscape, evolving business needs, as well as global regulatory requirements.
Our cybersecurity risk mitigation involves a range of threat defense and protection measures such as monitoring of systems, threat containment methods, penetration testing, conducting crises simulations, identity and access management, vulnerability scanning, promoting security and privacy awareness training to our global employees, improving internal processes and following a system of controls, including but not limited to back-up protocols, system restoration processes, and end-point protection on Company devices.
We seek to align our cybersecurity risk management with the NIST Cyber Security Framework, as well as industry best practices. Our cybersecurity incident response processes guide the detection, response and recovery from cybersecurity incidents and compliance with regulatory reporting requirements.
Governance
Board of Directors
Management
38
The Company’s CISO oversees the Company’s cybersecurity incident response plan and related processes that are designed to assess and manage material risks from cybersecurity threats. The Company’s CISO also coordinates with the Company’s Corporate & Legal Affairs team and third parties, such as consultants and legal advisors, to assess and manage material risks from cybersecurity threats. The Company’s CISO is informed about and monitors the prevention, detection, mitigation, and remediation of cybersecurity incidents pursuant to criteria set forth in the Company’s incident response plan and related processes.
Item 2. Properties
Solventum is a global organization with major centers throughout the world, including the license of the Company's U.S. principal office located at 3M Center in Maplewood, Minnesota from 3M Company. Additionally, the Company owns, leases or otherwise has rights to use a number of facilities, including administration, research and development, manufacturing, warehousing, distribution and other facilities across the United States and globally. The Company owns, leases or otherwise has rights to use approximately 270 facilities consisting of 20 owned facilities and approximately 250 facilities that are leased or otherwise have rights to use. The Company has 7 manufacturing facilities in the United States and 14 outside of the United States, including Canada, China, Germany, Ireland, Mexico, and Poland. Many of these facilities will serve more than one of our business segments and multiple functions across our business.
The Company's facilities are well-maintained and adequate for current needs. Solventum continues to evaluate its facility needs as the Company progresses on its journey as a separate company.
Item 3. Legal Proceedings
Discussion of legal matters is incorporated by reference from Part II, Item 8, Note 12, "Commitments and Contingencies," to the consolidated financial statements, and should be considered an integral part of Part I, Item 3, "Legal Proceedings."
Item 4. Mine Safety Disclosures
Not applicable.
39
SOLVENTUM CORPORATION
ANNUAL REPORT ON FORM 10-K
For the Year Ended December 31, 2025
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
At February 18, 2026, there were 43,602 shareholders of record. Solventum’s stock ticker symbol is SOLV and is listed on the New York Stock Exchange. No cash dividends were declared or paid since the company's inception on April 1, 2024.
Issuer Purchases of Equity Securities
In November 2025, Solventum's Board of Directors approved a share repurchase program, which authorizes the Company to purchase up to $1 billion of the Company's outstanding common stock.
Pursuant to the authorization, repurchases may be made from time to time in the open market, including through a Rule 10b5-1 or Rule 10b-18 trading plan. The share repurchase program has no time limit, does not require the Company to acquire any amount of shares, and may be suspended or discontinued at any time.
There were no issuer purchases of equity securities under the share repurchase program for the three months ended December 31, 2025.
Stock Performance Graph
The following graph compares the total shareholder return on the Company's common stock for the last 21 months since the company started trading on April 1, 2024 with the Standard & Poor's ("S&P") 500 and S&P 500 Healthcare indices. The graph assumes $100 was invested in each of these indices on April 1, 2024, the first day of "regular way" trading, for the Company's common stock. The comparisons are based on historical data and are not indicative of, nor intended to forecast, the future performance of the Company's common stock.
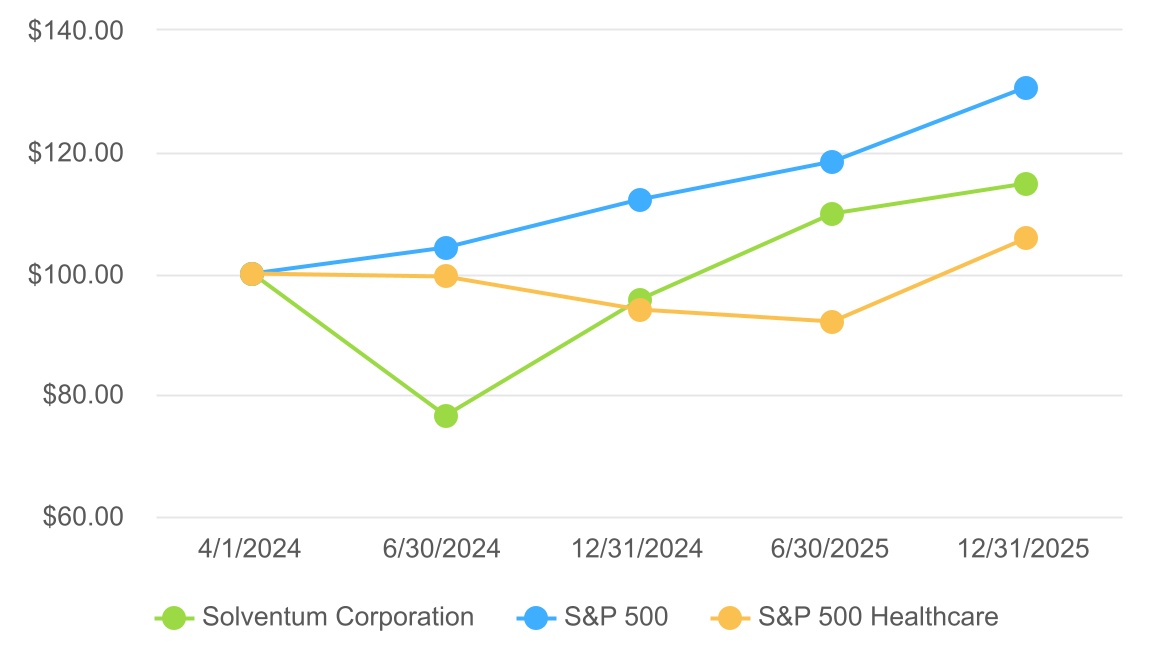
| Company/Index | April 1, 2024 | June 30, 2024 | December 31, 2024 | June 30, 2025 | December 31, 2025 | |||||||||||||||
| Solventum Corporation | $ | 100.00 | $ | 76.53 | $ | 95.60 | $ | 109.75 | $ | 114.67 | ||||||||||
| S&P 500 Index | 100.00 | 104.13 | 112.16 | 118.33 | 130.55 | |||||||||||||||
| S&P 500 Health Care Index | 100.00 | 99.50 | 93.91 | 92.02 | 105.68 | |||||||||||||||
Item 6. [Reserved]
40
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with the Company’s consolidated financial statements and corresponding notes elsewhere in this Annual Report on Form 10-K. The following discussion and analysis provides information management believes to be relevant to understanding the financial condition and results of operations of Solventum for the years ended December 31, 2025 and 2024. Discussion, analysis and comparisons of the year ended December 31, 2023 that are not included in this Annual Report on Form 10-K can be found in "Management's Discussion and Analysis of Financial Condition and Results of Operations" in the Annual Report on Form 10-K for the year ended December 31, 2024 filed on February 28, 2025. This discussion contains forward-looking statements that are based upon current expectations and are subject to uncertainty and changes in circumstances. Our actual results could differ materially from the results contemplated by these forward-looking statements due to a number of factors, including those discussed below and elsewhere in this Annual Report on Form 10-K, particularly in "Risk Factors." See "Cautionary Note Regarding Forward-Looking Statements."
All amounts discussed are in millions of U.S. dollars, unless otherwise indicated. Amounts reported within this Annual Report are rounded to the nearest million and the sum of the components may not equal the total amount reported due to rounding. Additionally, certain columns and rows within tables may not sum due to rounding.
Unless the context otherwise requires, references to "Solventum" and the "Company" refer to (i) 3M’s Health Care Business prior to the Spin-Off as a carve-out business of 3M and (ii) Solventum Corporation and its subsidiaries following the Spin-Off.
Transition to Standalone Company
Solventum utilized allocations and carve-out methodologies through the date of the Spin-Off to prepare combined financial statements. The consolidated financial statements herein for periods prior to the Spin-Off may not be indicative of the Company’s future performance, do not necessarily include the actual expenses that would have been incurred, and may not reflect our results of operations, financial position, and cash flows had we been a separate, standalone company during the historical periods presented.
In particular, Solventum benefited from 3M’s long operating history, reputation and well-known brand. Following the separation, Solventum is operating under its own brand, and accordingly may be negatively impacted due to the loss of benefits conferred by 3M’s brand recognition and reputation. In addition, the debt obligations incurred by Solventum in connection with the separation will adversely affect its profitability and could affect its ability to use its cash flow for investing in the business, strategic transactions, including mergers and acquisitions, and returning capital. See Note 1, "Significant Accounting Policies - Organization and Description of Business and Basis of Presentation" to the consolidated financial statements and Part 1, Item 1A "Risk Factors" for additional information.
Management’s Discussion and Analysis of Financial Condition and Results of Operations ("MD&A") is designed to provide a reader of Solventum’s financial statements with a narrative from the perspective of management. Solventum’s MD&A is presented in the following sections:
•Overview
•Results of Operations
•Performance by Business Segment
•Geographic Area Supplemental Information
•Critical Accounting Estimates
•New Accounting Pronouncements
•Financial Condition and Liquidity
•Financial Instruments
Overview
Solventum is a leading global healthcare company developing, manufacturing, and commercializing a broad portfolio of solutions that leverages deep material science, data science, and digital capabilities to address critical customer and patient needs. We constantly seek to enable the improvement of standards of care and move healthcare forward with innovation powered by insights, clinical intelligence, technology, and manufacturing expertise. Our 70+ year history of discovering and innovating advanced solutions has helped us solve our customers’ toughest challenges and become a trusted partner.
41
Operating Segments and Sales Change Information
Solventum manages its operations in three reportable business segments: MedSurg, Dental Solutions, and Health Information Systems. On February 25, 2025, the Company entered into a Transaction Agreement to sell its Purification and Filtration business to Thermo Fisher Scientific Inc. ("Buyer"). On June 25, 2025, the Company and Buyer entered into an Amended and Restated Transaction Agreement to exclude the Company’s drinking water filtration business (the "Water Business") from the scope of the Purification and Filtration business to be acquired by Buyer (such acquired business, the "Business"). On September 1, 2025, Solventum completed the sale of the Business to the Buyer in accordance with the terms of the Agreement. The cash consideration paid to Solventum at closing was approximately $4 billion. Refer to Note 3, "Acquisitions and Divestitures" for additional information.
References are made to organic sales change, which is defined as the change in net sales, absent the separate impacts on sales from foreign currency translation and acquisitions, net of divestitures. Constant currency, as reflected in the tables below, is defined as the change in net sales absent the impact on sales from foreign currency translation. Other, as comprised in the tables below, includes acquisition and divestiture-related activities. Acquisitions include sales from Acera that was acquired in December 2025, non-health care related supply agreements that conveyed from 3M to the Company at Spin-Off and sales from new supply agreements with 3M that commenced at Spin-Off. Divestiture impacts include lost sales from the Company’s Purification and Filtration business that was sold in September 2025, certain health care businesses retained by 3M India in connection with the Spin-Off, as well as impacts from other immaterial divested businesses. Solventum believes this information is useful to investors and management in understanding ongoing operations and in analysis of ongoing operating trends.
Healthcare Market Drivers
Changing demographics
An aging population, the prevalence and incidence rates of chronic conditions, and a rising middle class are driving the demand for improved access to quality care.
Optimizing workflows to improve care quality and operational efficiency
Of the $5.3 trillion in annual U.S. healthcare spending, an estimated 25% represents administrative costs that do not contribute to health outcomes and which we believe to be potentially wasteful based on overall spending data reported by the Centers for Medicare & Medicaid Services in the NHE Fact Sheet (available on CMS.gov as of January 14, 2026) and administrative spending estimates published in JAMA (Shrank et. al., Waste in the US Health Care System: Estimated Costs and Potential for Savings, published October 7, 2019). As healthcare providers and payers face increasing reimbursement constraints and evolving payment models, the need to reduce avoidable administrative costs has become more acute. Our solutions are designed to optimize workflows, enabling clinicians to be more productive by spending less time on administrative tasks and more time focused on improving the patient care experience. Our solutions also support reducing infections and complications that lead to an increase in avoidable administrative and clinical costs.
Increasing digital technology and data-driven care delivery
Both clinicians and patients have shifted their preferences towards utilizing digitally enabled solutions to provide data-driven care. Whether it is interactions with patients through a digital interface or the use of data, analytics, and artificial intelligence (AI) to support informed health decisions, the need for digital tools in the healthcare industry has grown over time. Our solutions integrate digital processes, AI-enabled capabilities and data in multiple ways and across different parts of the healthcare industry and are intended to enable efficient and effective delivery of care.
Shifting care from the hospital to lower-cost care sites
Although hospitals continue to be a core site for delivery of care, patients are increasingly looking for flexibility of care when and where they need it. Alternative care sites, such as ambulatory surgery centers, wound care clinics, retail pharmacies, and the home, are more affordable and accessible to patients. We believe our solutions enable clinicians to extend their care delivery from acute to ambulatory to home settings without compromising the quality of care and while reducing the total cost of care.
Increasing demand for personalized care
Engaging patients in a personalized way allows clinicians to provide a better care experience while improving outcomes and reducing costs. This spans several areas of healthcare, including customized orthodontic aligner treatments, and follow-up wound care at home. We believe our solutions deliver personalized care options in a way that is patient-centric, scalable, and cost-effective.
42
Our ability to take advantage of these market opportunities will be subject to various risks, including general economic, business and market dynamic risks, the impact of our separation from 3M; and the cost to service the debt we incurred in connection with the separation. See Part I, Item 1A, "Risk Factors" in this Annual Report on Form 10-K, for a discussion of these risks, which you should consider carefully.
Sales and operating income by business segment:
The following tables contain sales and operating results by business segment for all periods presented. Refer to the section entitled "—Performance by Business Segment" below for discussion of sales change and operating performance. Refer to Note 18 to the consolidated financial statements for additional information on the Company's business segments.
Segment and Total Company Net Sales
| Year ended December 31, | ||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | Reported Growth | Currency Impact | Constant Currency | Other | Organic Growth | |||||||||||||||||||||||||||||||||||||
| Segment Sales | ||||||||||||||||||||||||||||||||||||||||||||
| Advanced Wound Care | $ | 1,883 | $ | 1,835 | 2.6 | % | 0.5 | % | 2.1 | % | 0.1 | % | 2.0 | % | ||||||||||||||||||||||||||||||
| Infection Prevention and Surgical Solutions | 2,934 | 2,802 | 4.7 | 0.5 | 4.2 | (0.4) | 4.5 | |||||||||||||||||||||||||||||||||||||
| MedSurg | 4,817 | 4,637 | 3.9 | 0.6 | 3.3 | (0.2) | 3.5 | |||||||||||||||||||||||||||||||||||||
| Dental Solutions | 1,349 | 1,295 | 4.2 | 1.1 | 3.1 | (0.2) | 3.3 | |||||||||||||||||||||||||||||||||||||
| Health Information Systems | 1,360 | 1,306 | 4.1 | 0.2 | 3.9 | — | 4.0 | |||||||||||||||||||||||||||||||||||||
| Purification and Filtration | 497 | 709 | (29.9) | 1.1 | (31.0) | (36.5) | 5.5 | |||||||||||||||||||||||||||||||||||||
| All Other | 302 | 306 | (1.5) | 0.3 | (1.8) | 4.3 | (6.1) | |||||||||||||||||||||||||||||||||||||
| Total Company | $ | 8,325 | $ | 8,254 | 0.9 | % | 0.6 | % | 0.3 | % | (3.0) | % | 3.3 | % | ||||||||||||||||||||||||||||||
| Year ended December 31, | ||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | 2024 | 2023 | Reported Growth | Currency Impact | Constant Currency | Other | Organic Growth | |||||||||||||||||||||||||||||||||||||
| Segment Sales | ||||||||||||||||||||||||||||||||||||||||||||
| Advanced Wound Care | $ | 1,835 | $ | 1,826 | 0.5 | % | (0.3) | % | 0.8 | % | (0.1) | % | 0.9 | % | ||||||||||||||||||||||||||||||
| Infection Prevention and Surgical Solutions | 2,802 | 2,805 | (0.1) | (0.8) | 0.7 | (0.7) | 1.4 | |||||||||||||||||||||||||||||||||||||
| MedSurg | 4,637 | 4,632 | 0.1 | (0.6) | 0.7 | (0.5) | 1.2 | |||||||||||||||||||||||||||||||||||||
| Dental Solutions | 1,295 | 1,329 | (2.6) | (0.7) | (2.0) | (1.5) | (0.4) | |||||||||||||||||||||||||||||||||||||
| Health Information Systems | 1,306 | 1,285 | 1.6 | — | 1.6 | — | 1.6 | |||||||||||||||||||||||||||||||||||||
| Purification and Filtration | 709 | 689 | 3.0 | (0.6) | 3.7 | (0.9) | 4.6 | |||||||||||||||||||||||||||||||||||||
| All Other | 306 | 262 | 16.7 | (0.9) | 17.6 | 18.8 | (1.2) | |||||||||||||||||||||||||||||||||||||
| Total Company | $ | 8,254 | $ | 8,197 | 0.7 | % | (0.5) | % | 1.2 | % | — | % | 1.2 | % | ||||||||||||||||||||||||||||||
43
Segment and Total Company Operating Income
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2025 vs 2024 change | |||||||||||||||||
| Segment Operating Income | ||||||||||||||||||||
| MedSurg | $ | 810 | $ | 887 | (8.6) | % | ||||||||||||||
| Dental Solutions | 346 | 350 | (1.1) | |||||||||||||||||
| Health Information Systems | 496 | 431 | 15.0 | |||||||||||||||||
| Purification and Filtration | 96 | 74 | 29.7 | |||||||||||||||||
| All Other | 42 | 30 | 40.0 | |||||||||||||||||
| Corporate and Unallocated | 390 | (736) | 153.0 | |||||||||||||||||
| Total Company | $ | 2,181 | $ | 1,036 | 110.5 | % | ||||||||||||||
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2024 | 2023 | 2024 vs 2023 change | |||||||||||||||||
| Segment Operating Income | ||||||||||||||||||||
| MedSurg | $ | 887 | $ | 1,107 | (19.9) | % | ||||||||||||||
| Dental Solutions | 350 | 442 | (20.8) | |||||||||||||||||
| Health Information Systems | 431 | 423 | 1.9 | |||||||||||||||||
| Purification and Filtration | 74 | 111 | (33.3) | |||||||||||||||||
| All Other | 30 | 51 | (41.2) | |||||||||||||||||
| Corporate and Unallocated | (736) | (442) | 66.5 | |||||||||||||||||
| Total Company | $ | 1,036 | $ | 1,692 | (38.8) | % | ||||||||||||||
44
Net Sales by Geographic Area
While the Company manages its businesses globally and believes its business segment results are the most relevant measure of performance, the Company also utilizes geographic area data as a secondary performance measure. Sales are generally reported within the geographic area based on the location of the customer taking possession of the products or in which services are rendered. Additional geographic financial information related to the Company’s operations is provided in Note 18 in the accompanying consolidated financial statements.
Percent change information compares year ended December 31, 2025 and December 31, 2024 with the same periods for the prior year, unless otherwise indicated.
| Year ended December 31, 2025 | ||||||||||||||||||||
| (Millions) | United States | International | Worldwide | |||||||||||||||||
| Net sales | $ | 4,668 | $ | 3,657 | $ | 8,325 | ||||||||||||||
| % of worldwide sales | 56.1 | % | 43.9 | % | 100.0 | % | ||||||||||||||
| Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 3.7 | % | 2.7 | % | 3.3 | % | ||||||||||||||
| Other | (1.3) | (5.1) | (3.0) | |||||||||||||||||
| Constant Currency | 2.4 | (2.4) | 0.3 | |||||||||||||||||
| Currency Impact | — | 1.4 | 0.6 | |||||||||||||||||
| Reported Growth | 2.4 | % | (1.0) | % | 0.9 | % | ||||||||||||||
| Year ended December 31, 2024 | ||||||||||||||||||||
| (Millions) | United States | International | Worldwide | |||||||||||||||||
| Net sales | $ | 4,559 | $ | 3,695 | $ | 8,254 | ||||||||||||||
| % of worldwide sales | 55.2 | % | 44.8 | % | 100.0 | % | ||||||||||||||
| Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 1.2 | % | 1.4 | % | 1.2 | % | ||||||||||||||
| Other | 0.8 | (1.0) | — | |||||||||||||||||
| Constant Currency | 2.0 | 0.4 | 1.2 | |||||||||||||||||
| Currency Impact | — | (1.2) | (0.5) | |||||||||||||||||
| Reported Growth | 2.0 | % | (0.8) | % | 0.7 | % | ||||||||||||||
Additional information beyond what is included in the preceding table is as follows:
Year ended 2025 results
•In the United States geographic area, both total sales and organic sales increased. Organic growth was led by MedSurg and Health Information Systems. Other is comprised of lost sales due to the divestiture of the Purification and Filtration business in September 2025.
•In the International geographic area, total sales declined while organic sales increased. Organic growth was led by MedSurg and Dental Solutions. Other is comprised of lost sales due to the divestiture of the Purification and Filtration business in September 2025.
Year ended 2024 results
•In the United States geographic area, both total sales and organic sales increased. Organic growth was led by MedSurg and Health Information Systems.
•In the International geographic area, total sales decreased while organic sales increased. Organic growth was led by MedSurg and Purification and Filtration.
Managing currency risks
Prior to April 1, 2024, Solventum indirectly participated in 3M’s centrally managed hedging program. Starting in the second quarter of 2024, Solventum established its own hedging program. Refer to Note 11 to the consolidated financial statements for additional details.
45
Foreign currency had a positive worldwide impact on sales for the year ended December 31, 2025 compared to 2024. Solventum estimates that year-on-year foreign currency transaction effects, including hedging impacts, increased pre-tax income by approximately $6 million in 2025.
The stronger U.S. dollar had a negative worldwide impact on sales for the year ended December 31, 2024 compared to 2023. Solventum estimates that year-on-year foreign currency transaction effects, including hedging impacts, decreased pre-tax income by approximately $23 million in 2024.
Financial condition
Refer to the section entitled "—Financial Condition and Liquidity" below for a discussion of items impacting cash flows.
Results of Operations
Net Sales
Refer to the preceding "—Overview" section and the "—Performance by Business Segment" section later in MD&A for discussion of sales change.
Costs of Sales
| Year ended Year ended December 31, | ||||||||||||||||||||||||||||||||
| (Percent of corresponding net sales) | 2025 | 2024 | 2023 | 2025 vs 2024 | 2024 vs 2023 | |||||||||||||||||||||||||||
| Cost of product | 53.6 | % | 50.0 | % | 48.0 | % | 3.6 | % | 2.0 | % | ||||||||||||||||||||||
| Cost of software and rentals | 23.9 | 25.7 | 25.3 | (1.8) | 0.4 | |||||||||||||||||||||||||||
Costs of Product
Costs of product includes manufacturing, engineering and logistics costs. The Company operates a global supply chain and sourcing organization, including product sourced under master supply and transition manufacturing agreements with 3M. As a result, the Company is impacted by changes in the global regulatory and economic environment, including tariffs. The evolving regulatory and economic environment may impact our cost or ability to source products. To the extent possible the Company takes actions to offset these costs or identify alternative sources of supply.
Costs of product, measured as a percent of sales of product, increased in 2025 when compared to 2024. The increase was driven by the impact of new tariffs of approximately $55 million, the full year impact from inventory sourced under the master supply and transition manufacturing agreements with 3M, and higher logistics costs, partially offset by benefits from cost savings programs.
Costs of product, measured as a percent of sales of product, increased in 2024 when compared to 2023. The increase was driven by increased costs due to the impact of higher costs on inventory sourced under the master supply and transition manufacturing agreements with 3M and due to the cost of other transition support provided by 3M that have been incurred since Spin-Off.
Costs of Software and Rentals
Costs of software and rentals includes compensation-related costs associated with installation, training and maintenance for our software products, and depreciation, maintenance and refurbishment costs and logistics costs related to our hardware rental units.
Costs of software and rentals, measured as a percent of sales of software and rentals, decreased in 2025 as compared to 2024. The decrease was due to the impact of lower external license fees, price and sales mix, primarily driven by higher sales of our revenue cycle management solution.
Costs of software and rentals, measured as a percent of sales of software and rentals, increased in 2024 as compared to 2023. This increase was driven by higher compensation costs.
46
Operating Expenses
| Year ended December 31, | ||||||||||||||||||||||||||||||||
| (Percent of total net sales) | 2025 | 2024 | 2023 | 2025 vs 2024 | 2024 vs 2023 | |||||||||||||||||||||||||||
| Selling, general and administrative (SG&A) | 37.0 | % | 33.7 | % | 28.0 | % | 3.3 | % | 5.7 | % | ||||||||||||||||||||||
| Research and development (R&D) | 8.9 | 9.4 | 9.2 | (0.5) | 0.2 | |||||||||||||||||||||||||||
| Gain on sale of business | 18.6 | — | 0.7 | 18.6 | (0.7) | |||||||||||||||||||||||||||
| Operating Income | 26.2 | 12.6 | 20.6 | 13.6 | (8.0) | |||||||||||||||||||||||||||
Selling, General and Administrative
SG&A, measured as a percent of total net sales, increased in 2025 when compared to 2024. The increase was driven by costs incurred to separate the Purification and Filtration business, higher compensation, including equity-based awards, and higher costs associated with both initial stand-up and ongoing operations to support a standalone company.
SG&A, measured as a percent of total net sales, increased in 2024 when compared to 2023. The increase was driven by higher compensation, including equity-based awards, and higher costs associated with both initial stand-up and ongoing operations to support a standalone company.
Research and Development
R&D, measured as a percent of total net sales, decreased slightly in 2025 when compared to 2024 primarily due to reimbursement of a portion of our technical development costs from 3M related to our supply chain separation.
R&D, measured as a percent of total net sales, increased slightly in 2024 when compared to 2023 due to initial stand-up costs. The Company continues to prioritize investment initiatives.
Gain on Sale of Business
The gain on sale of business primarily relates to the Company's completed sale of the Purification and Filtration business in the third quarter of 2025, which resulted in a net gain of $1.5 billion for 2025.
Interest Expense, Net, Loss on Debt Extinguishment, Net, and Other Expense (Income), Net
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Interest expense, net | $ | 347 | $ | 367 | $ | — | ||||||||||||||
| Loss on debt extinguishment, net | 82 | — | — | |||||||||||||||||
| Other expense (income), net | 39 | 64 | 25 | |||||||||||||||||
Interest expense, net includes interest accrued on debt obligations, offset by interest income from cash and marketable securities. Interest expense, net decreased in 2025 as compared to 2024 due to lower interest expense as a result of lower debt outstanding. Interest expense, net increased in 2024 as compared to 2023 due to interest incurred on the February 2024 issuance of senior notes and March 2024 draw on the senior term loan credit facilities. Refer to Note 9 to the consolidated financial statements for more information. This increase was partially offset by interest earned from cash and marketable securities held during the period.
Loss on debt extinguishment, net includes charges incurred in the third quarter of 2025 from the differential between carrying value and the amount paid to acquire the tendered Senior Notes and related expenses. Refer to Note 9 to the consolidated financial statements for additional information. These charges were partially offset by the gain from interest rate swaps entered into and subsequently settled in connection with the sale of the Purification and Filtration business. Refer to Note 11 to the consolidated financial statements for additional information.
Other expense (income), net includes the non-service component of periodic pension cost, investment gains and losses, and foreign currency transaction gain (loss). Other expense (income), net decreased in 2025 as compared to 2024 primarily due to charges associated with the substantial liquidation of foreign operations completed as part of our separation from 3M. Other expense (income), net increased in 2024 as compared to 2023 resulting from charges associated with the substantial liquidation of foreign operations completed as part of our separation from 3M in addition to foreign currency impacts and investment losses.
47
Provision (benefit) for Income Taxes:
| Year ended December 31, | ||||||||||||||||||||
| (Percent of pre-tax income/loss) | 2025 | 2024 | 2023 | |||||||||||||||||
| Effective tax rate | 9.2 | % | 20.9 | % | 19.3 | % | ||||||||||||||
The change in the effective tax rate in 2025 when compared to 2024 was primarily driven by impacts associated with the sale of the Purification and Filtration business. Refer to Note 8 to the consolidated financial statements for additional detail on the Company's effective tax rate.
Performance by Business Segment
Note 18 to the consolidated financial statements provides an overview of Solventum’s reportable business segments. Upon closing the sale of our Purification and Filtration business, we primarily manage our operations in three business segments: MedSurg, Dental Solutions, and Health Information Systems. Our Chief Operating Decision Maker evaluates segment operating performance using net sales and business segment operating income.
All Other
All Other primarily consists of the Water Business that was retained after the sale of the Purification and Filtration Business. All Other also includes sales and cost of sales related to our agreements to supply 3M and other supply agreements assumed by the Company at Spin-Off related to legacy 3M businesses, which were historically included within Corporate and Unallocated.
Corporate and Unallocated
Certain items are maintained at the corporate level and not allocated to the segments ("Corporate and Unallocated"). Corporate and Unallocated primarily includes amortization of acquired intangible assets, restructuring and related charges, timing related benefits or costs associated with capitalized manufacturing variances, charges and recoveries related to certain litigation, transaction and employee retention costs related to the acquisition of Acera, and gains on sale of businesses. In addition, Corporate and Unallocated includes Spin-Off and separation related costs. Spin-Off and separation related costs include any costs incurred as part of our separation from 3M and costs to setup operations as a standalone company, including system implementations, manufacturing relocations, legal entity separations, certain equity awards granted as part of the Spin-Off, profit mark-ups on transition service arrangements with 3M and other one-time costs. Corporate and Unallocated also includes income and costs related to transition service agreements entered into in connection with the sale of the Purification and Filtration business.
Because Corporate and Unallocated includes a variety of miscellaneous items, it is subject to fluctuation on a quarterly and annual basis.
Operating Business Segments
Information related to the Company’s segments is presented in the tables that follow with additional context in the corresponding narrative below the tables.
MedSurg (57.9% of consolidated sales for the year ended December 31, 2025 )
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net sales | $ | 4,817 | $ | 4,637 | $ | 4,632 | ||||||||||||||
Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 3.5 | % | 1.2 | % | ||||||||||||||||
Other | (0.2) | (0.5) | ||||||||||||||||||
| Constant currency | 3.3 | 0.7 | ||||||||||||||||||
| Currency impact | 0.6 | (0.6) | ||||||||||||||||||
| Reported growth | 3.9 | % | 0.1 | % | ||||||||||||||||
Business segment operating income (millions) | $ | 810 | $ | 887 | $ | 1,107 | ||||||||||||||
| Percent change | (8.6) | % | (19.9) | % | ||||||||||||||||
| Percent of sales | 16.8 | % | 19.1 % | 23.9 % | ||||||||||||||||
48
Year 2025 results:
Sales in MedSurg were up 3.9%:
•Organic growth was driven by volumes in our Infection Prevention and Surgical Solutions business, led by I.V. site management. Growth within our Advanced Wound Care business was led by volume growth in negative pressure wound therapy.
•Other primarily includes lost sales from certain health care businesses retained by 3M India in connection with the Spin-Off.
•Foreign currency translation positively impacted sales by 0.6%.
Business segment operating income margin decreased when compared to the same period last year. The decrease was primarily driven by the impact of higher product costs due to tariffs, logistics and a full year of supply agreement mark-ups from 3M.
Year 2024 results:
Sales in MedSurg were up 0.1%:
•Organic sales growth of 1.2% was driven by volumes, primarily due to benefits from medical OEM products, I.V. site management, and single-use negative pressure wound therapy, partially offset by declines in traditional negative pressure wound therapy and sterilization assurance products.
•Other includes lost sales from certain health care businesses retained by 3M India in connection with the Spin-Off.
•Foreign currency translation negatively impacted sales by (0.6%).
Business segment operating income margin decreased when compared to the same period last year. The decrease was driven by higher costs to stand-up and operate our standalone structure after Spin-Off.
Dental Solutions (16.2% of consolidated sales for the year ended December 31, 2025)
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net sales | $ | 1,349 | $ | 1,295 | $ | 1,329 | ||||||||||||||
Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 3.3 | % | (0.4) | % | ||||||||||||||||
Other | (0.2) | (1.5) | ||||||||||||||||||
| Constant currency | 3.1 | (2.0) | ||||||||||||||||||
| Currency impact | 1.1 | (0.7) | ||||||||||||||||||
| Reported growth | 4.2 | % | (2.6) | % | ||||||||||||||||
| Business segment operating income | $ | 346 | $ | 350 | $ | 442 | ||||||||||||||
| Percent change | (1.1) | % | (20.8) | % | ||||||||||||||||
| Percent of sales | 25.6 | % | 27.0 | % | 33.3 | % | ||||||||||||||
Year 2025 results:
Sales in Dental Solutions were up 4.2%:
•Organic growth was primarily driven by new product volume growth in restorative and prevention solutions, partially offset by a decline in traditional orthodontic products.
•Other is driven by lost sales from certain health care businesses retained by 3M India in connection with the Spin-Off.
•Foreign currency translation positively impacted sales by 1.1%.
Business segment operating income margin decreased when compared to the same period last year as a result of higher logistics costs, tariffs and other costs to operate our standalone structure after Spin-Off.
49
Year 2024 results:
Sales in Dental Solutions were down (2.6%):
•Volume declines associated with softening end-market demand were partially offset by the favorable impact of prior year price actions.
•Other is primarily driven by lost sales from the Company’s dental anesthetics business that was sold in August 2023 as well as lost sales from certain health care businesses retained by 3M India in connection with the Spin-Off.
•Foreign currency translation negatively impacted sales by (0.7%).
Business segment operating income margin decreased when compared to the same period last year as a result of higher costs to stand-up and operate our standalone structure after Spin-Off.
Health Information Systems (16.3% of consolidated sales for year ended December 31, 2025)
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net sales | $ | 1,360 | $ | 1,306 | $ | 1,285 | ||||||||||||||
Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 4.0 | % | 1.6 | % | ||||||||||||||||
Other | — | — | ||||||||||||||||||
| Constant currency | 3.9 | 1.6 | ||||||||||||||||||
| Currency impact | 0.2 | — | ||||||||||||||||||
| Reported growth | 4.1 | % | 1.6 | % | ||||||||||||||||
| Business segment operating income | $ | 496 | $ | 431 | $ | 423 | ||||||||||||||
| Percent change | 15.0 | % | 1.9 | % | ||||||||||||||||
| Percent of sales | 36.5 | % | 33.0 | % | 32.9 | % | ||||||||||||||
Year 2025 results:
Sales in Health Information Systems were up 4.1%:
•Positive organic growth was driven by expanded adoption of our Solventum™ 360 EncompassTM and performance management solutions.
•Clinician productivity solutions declined primarily due to impacts from changing market conditions.
•Foreign currency translation positively impacted sales by 0.2%.
Business segment operating income margin increased when compared to the same period last year, driven by sales price growth, product mix and lower external license fees.
Year 2024 results:
Sales in Health Information Systems were up 1.6%:
•Positive sales growth was driven by continued adoption of our 3MTM 360 EncompassTM.
•Clinician productivity solutions declined primarily due to impacts from changing market conditions.
Business segment operating income margin increased slightly when compared to the same period last year as product mix benefit due to higher software sales and lower professional services was partially offset by higher compensation costs.
50
Purification and Filtration (6.0% of consolidated sales for the year ended December 31, 2025)
| Year ended December 31, | ||||||||||||||||||||
| 2025 | 2024 | 2023 | ||||||||||||||||||
Net sales (millions) | $ | 497 | $ | 709 | $ | 689 | ||||||||||||||
Increase/(decrease) | ||||||||||||||||||||
| Organic growth | 5.5 | % | 4.6 | % | ||||||||||||||||
Other | (36.5) | (0.9) | ||||||||||||||||||
| Constant currency | (31.0) | 3.7 | ||||||||||||||||||
| Currency impact | 1.1 | (0.6) | ||||||||||||||||||
| Reported growth | (29.9) | % | 3.0 | % | ||||||||||||||||
| Business segment operating income (millions) | $ | 96 | $ | 74 | $ | 111 | ||||||||||||||
| Percent change | 29.7 | % | (33.3) | % | ||||||||||||||||
| Percent of sales | 19.3 | % | 10.4 | % | 16.1 | % | ||||||||||||||
Year 2025 results:
Sales in Purification and Filtration were down (29.9)%:
•Organic growth was driven by growth in both bioprocessing filtration and industrial filtration, which benefited from added production capacity, partially offset by declines in membrane OEM products.
•Other is driven by lost sales after the business was sold in September 2025.
•Foreign currency translation positively impacted sales by 1.1%.
Business segment operating income margin increased due to volume growth, favorable sales mix and a benefit resulting from the Company stopping depreciation on assets classified as held for sale, partially offset by higher costs to operate our standalone structure after Spin-Off.
Year 2024 results:
Sales in Purification and Filtration were up 3.0%:
•Primarily driven by higher volume growth in our bioprocessing filtration product category. This growth was partially offset by our membranes OEM.
•Other includes lost sales from certain health care businesses retained by 3M India in connection with the Spin-Off.
•Foreign currency translation negatively impacted sales by (0.6)%.
Business segment operating income margin decreased primarily due to the negative impact from costs to stand-up and operate our standalone structure after Spin-Off.
Geographic Area Supplemental Information
| Employees as of December 31, | Capital Spending for the year ended December 31, | Property, Plant and Equipment - net as of December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Millions, except Employees) | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | |||||||||||||||||||||||||||||||||||||||||||||||
| United States | 10,308 | 10,919 | 10,906 | $ | 200 | $ | 229 | $ | 160 | $ | 773 | $ | 893 | $ | 770 | |||||||||||||||||||||||||||||||||||||||||
| International | 10,276 | 11,062 | 11,101 | 179 | 151 | 130 | 553 | 729 | 687 | |||||||||||||||||||||||||||||||||||||||||||||||
| Total Company | 20,584 | 21,981 | 22,007 | $ | 379 | $ | 380 | $ | 290 | $ | 1,326 | $ | 1,622 | $ | 1,457 | |||||||||||||||||||||||||||||||||||||||||
Employment:
Employment decreased in 2025 when compared to 2024 and decreased slightly in 2024 when compared to 2023. The above table includes the impact of acquisitions, divestitures, and other actions.
51
Capital Spending and Property, Plant and Equipment - Net:
Investments in property, plant and equipment enable growth across many diverse markets, helping to meet product demand and increasing manufacturing efficiency. The Company is increasing its investment in manufacturing and sourcing capability in order to more closely align its production capability with its sales in major geographic areas in order to best serve its customers throughout the world with proprietary, automated, efficient, safe and sustainable processes. Capital spending is discussed in more detail below in the section entitled "—Cash Flows from Investing Activities."
Critical Accounting Estimates
Information regarding significant accounting policies is included in Note 1 of the accompanying consolidated financial statements. The preparation of financial statements in conformity with U.S. generally accepted accounting principles requires management to make certain estimates and assumptions. Such estimates and assumptions are subject to inherent uncertainties which may result in actual amounts differing from these estimates.
The Company considers the items below to be critical accounting estimates. Critical accounting estimates are those estimates made in accordance with generally accepted accounting principles that involve a significant level of estimation uncertainty and have had or are reasonably likely to have a material impact on the financial condition or results of operations of the Company.
Legal Proceedings
Assessments of lawsuits and claims can involve a series of complex judgments about future events, the outcomes of which are inherently uncertain, and can rely heavily on estimates and assumptions. The Company accrues an estimated liability for legal proceeding claims that are both probable and reasonably estimable in accordance with Accounting Standard Codification (ASC) 450, Contingencies. Please refer to the section entitled "Process for Disclosure and Recording of Liabilities Related to Legal Proceedings" (contained in "Legal Proceedings" in Note 12 to the accompanying consolidated financial statements) for additional information about such estimates.
Goodwill and Intangible Assets
The Company makes certain estimates and judgments in impairment assessments of goodwill. Goodwill is tested for impairment annually in the fourth quarter of each year, as further discussed below, and is tested between annual tests if an event occurs or circumstances change that would indicate the carrying amount may be impaired. The Company may assess qualitative factors for its reporting units to determine whether it is more likely than not that the fair value of a reporting unit is less than its carrying amount including goodwill. Alternatively, the Company may bypass this qualitative assessment and perform a quantitative goodwill impairment test.
Impairment testing for goodwill is done at a reporting unit level, with all goodwill assigned to a reporting unit. An impairment loss would be recognized when the carrying amount of the reporting unit’s net assets exceeds the estimated fair value of the reporting unit. The estimated fair value of a reporting unit is determined based on a market approach using comparable company information such as EBITDA (earnings before interest, taxes, depreciation and amortization) multiples. The Company also performs a discounted cash flow analysis for certain reporting units where the market approach indicates additional review is warranted. A discounted cash flow analysis involves key assumptions including projected sales, EBITDA margins, capital expenditures, and discount rates. Changes in reporting unit earnings, comparable company information, and expected future cash flows, as well as underlying market and overall economic conditions, among other factors, make these estimates subject to uncertainty. The Company did not perform a discounted cash flow analysis for any reporting unit for any period presented, as the market approach analysis resulted in sufficient headroom between the fair value and the carrying value for each of the Company's reporting units.
As of December 31, 2025, goodwill totaled approximately $5.7 billion. The Company has four reporting units that are assigned goodwill, with the MedSurg reporting unit accounting for approximately 74 percent of the goodwill balance. In connection with our annual testing in the fourth quarter of 2025, no qualitative indicators of impairment were identified for any of the Company's reporting units. For reporting units where quantitative testing was completed, the fair value exceeded the carrying value of the reporting unit by at least 65 percent. On December 23, 2025, subsequent to our annual impairment test, the Company completed the acquisition of Acera. Preliminary goodwill related to the acquisition was recognized and is included in the balance of our MedSurg reporting unit at December 31, 2025. The Company will continue to monitor its reporting units for any triggering events or other indicators of impairment.
The Company acquires intangible assets in connection with business combinations, primarily developed technology, trade names, and customer relationships. These acquired intangible assets are recorded at their estimated fair value as of the acquisition date. The fair values of acquired intangible assets are determined using information available on the acquisition date and are based on estimates and assumptions that management believes are reasonable. These estimates may include the amount
52
and timing of projected future cash flows attributable to each class of intangible asset, the discount rates used to present value those cash flows, and the assessment of the asset’s expected life cycle. Significant assumptions vary by the class of intangible asset and the valuation technique applied.
New Accounting Pronouncements
Information regarding new accounting pronouncements is included in Note 1 to the Company's consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
Financial Condition and Liquidity
Solventum's principal sources of liquidity are our existing cash and cash equivalents, cash generated from operations, and access to both our revolving credit facility and commercial paper program, which the Company believes will satisfy our foreseeable operating needs, capital expenditures, and debt service requirements. Discretionary cash may be allocated to strategic acquisitions, share repurchases, or repayment of debt obligations. The Company's cash position reflects business results and a global cash management strategy that leverages liquidity management along with analyzing economic factors and tax considerations.
Debt and Credit Facilities
Refer to Note 9 of the Company's consolidated financial statements included elsewhere in this Annual Report on Form 10-K for more information.
The Company had approximately $82 million in bank guarantees, surety bonds, and other similar instruments issued and outstanding at December 31, 2025. These instruments are utilized in connection with normal business activities.
Commercial Paper
On March 4, 2024, the Company entered into a commercial paper program that allows it to issue up to $2.0 billion aggregate principal amount of short-term notes to finance short-term liabilities. Any such issuance will mature within 364 days from date of issue. There was no commercial paper outstanding at December 31, 2025.
Cash, cash equivalents and marketable securities
As of December 31, 2025, Solventum had $878 million of cash and cash equivalents, of which approximately $800 million was held by the Company’s foreign subsidiaries and approximately $78 million was held in the United States. These balances are invested in bank instruments and other high-quality fixed income securities. There was an immaterial amount of marketable securities at December 31, 2025.
Cash Flows
Cash flows from operating, investing and financing activities are provided in the table that follows. Individual amounts in the consolidated statements of cash flows exclude the effect of exchange rate impacts on cash and cash equivalents, which are presented separately in the cash flows. Thus, the amounts presented in the following operating, investing and financing activities tables reflect changes in balances from period to period adjusted for these effects.
| Year Ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Cash provided by (used in): | ||||||||||||||||||||
| Operating activities | $ | 369 | $ | 1,185 | $ | 1,915 | ||||||||||||||
| Investing activities | 2,797 | (380) | (230) | |||||||||||||||||
| Financing activities | (3,057) | (240) | (1,552) | |||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | 7 | 3 | — | |||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | $ | 116 | $ | 568 | $ | 133 | ||||||||||||||
53
Operating Activities
In 2025, cash flows provided by operating activities decreased compared to 2024 primarily due to lower net income, excluding the gain on sale of the Purification and Filtration business, driven by higher costs to separate from 3M as well as higher transaction related activity including costs incurred in connection with closing the Purification and Filtration divestiture and higher receivables and other assets required to support the related transition service agreements.
In 2024, cash flows provided by operating activities decreased compared to 2023 primarily due to lower net income. Cash flow activity with 3M is reflected in the due from and due to related parties. This activity includes settlement of payables and receivables transferred at Spin-Off related to operating transactions between 3M and Solventum entities that occurred prior to the Spin-Off and transactions under the transition agreements with 3M.
Investing Activities
The increase in investing activities is related to proceeds from sale of the Purification and Filtration business in September 2025, partially offset by the payment to acquire Acera in December 2025. Purchases of property, plant and equipment remained flat in 2025 as compared to 2024. The company continues to focus capital spending on separation related activities as the Company relocates manufacturing and source of supply from 3M.
Purchases of property, plant and equipment increased in 2024 as compared to 2023. The increase is primarily driven by additional separation related capital spending as the Company relocates manufacturing and source of supply from 3M.
Financing Activities
Financing cash outflows increased in 2025 primarily due to the Company's repayment of $2.0 billion of senior notes via tender offers, including extinguishment costs, upon completion of the sale of the Purification and Filtration business and $870 million repayment of outstanding principal issued under the three year senior unsecured term loan credit facility. In addition, the Company repaid the remaining $200 million aggregate principal amount outstanding under the eighteen month senior unsecured term loan credit facility.
2024 proceeds from long-term debt of $8.3 billion were related to the first quarter issuance of $6.9 billion in senior notes and $1.5 billion in senior term loan credit facilities. The proceeds from these financing transactions were transferred to 3M in connection with the Spin-Off transaction, other than the amounts retained in order to achieve the $600 million retained cash target. During 2024, the Company repaid $300 million outstanding principal issued under the senior term loan credit facilities.
Material Cash Requirements from Known Contractual and Other Obligations:
Solventum’s material cash requirements from known contractual and other obligations primarily relate to the following, for which information on both a short-term and long-term basis is provided in the indicated notes to the consolidated financial statements:
•Tax obligations—Refer to Note 8 to the consolidated financial statements.
•Debt—Refer to Note 9 to the consolidated financial statements.
•Commitments and contingencies—Refer to Note 12 to the consolidated financial statements.
•Operating leases—Refer to Note 13 to the consolidated financial statements.
Solventum purchases the majority of its materials and services as needed, with no unconditional commitments. In limited circumstances, in the normal course of business, the Company enters into unconditional purchase obligations with various vendors that may take the form of, for example, take or pay contracts in which the Company guarantees payment to ensure availability of certain materials or services or to ensure ongoing efforts on capital projects. Additionally, the Company enters into contractual obligations for cloud storage solutions, enterprise resource planning and other IT-related services. The Company expects to receive underlying materials or services for these purchase obligations. To the extent these purchase obligations fluctuate, it largely trends with normal-course changes in regular operating activities. Additionally, contractual capital commitments represent a small part of the Company’s expected capital spending. As of December 31, 2025, unconditional purchase obligations aggregated to approximately $330 million over the next five years, primarily comprised of IT-related obligations.
54
Financial Instruments
The Company enters into foreign exchange forward contracts to hedge against the effect of exchange rate fluctuations on cash flows denominated in foreign currencies and to offset, in part, the impacts of changes in value of various non-functional currency denominated items including certain intercompany financing balances. As circumstances warrant, the Company also uses cross currency swaps as hedging instruments to hedge portions of the Company’s net investments in foreign operations. To help manage borrowing costs, the Company may enter into interest rate swaps, interest rate locks or other hedging instruments.
Refer to Item 7A, "Quantitative and Qualitative Disclosures About Market Risk," for further discussion of foreign exchange rates risk, and interest rates risk and commodity prices risk.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
In the context of Item 7A, Solventum is exposed to market risk due to the risk of loss arising from adverse changes in foreign currency exchange rates, interest rates and commodity prices. Changes in those factors could impact the Company’s results of operations and financial condition. The Company manages commodity price risks through negotiated supply contracts and price protection agreements. Senior management provides oversight for risk management and derivative activities, determines certain of the Company’s financial risk policies and objectives, and provides guidelines for derivative instrument utilization. Senior management also establishes certain associated procedures relative to control and valuation, risk analysis, counterparty credit approval, and ongoing monitoring and reporting.
To quantify Solventum’s primary market risk exposure, the Company performs a sensitivity analysis based on hypothetical changes in foreign currency spot exchange rates and interest rates as further described in the sections below. Note also that hypothetical changes in these rates were not applied to cash equivalents, accounts receivable, and accounts payable, because of the short-term nature of these instruments.
Refer to Note 1, "Significant Accounting Policies", Note 9, "Long-Term Debt and Short-Term Borrowings" and Note 11, "Derivatives" within the notes to the consolidated financial statements of this Annual Report on Form 10-K for additional discussion of foreign currency exchange, interest rates and financial instruments.
Foreign Currency Exchange Rates Risk: Foreign currency exchange rates and fluctuations in those rates may affect the Company’s net investment in foreign subsidiaries and may cause fluctuations in cash flows related to foreign denominated transactions. As circumstances warrant, the Company also uses cross-currency swap contracts as instruments to hedge portions of the Company’s net investments in foreign operations. Solventum is also exposed to the translation of foreign currency earnings to the U.S. dollar. The Company enters into foreign exchange forward contracts to hedge against the effect of exchange rate fluctuations on cash flows denominated in foreign currencies. These transactions are designated as cash flow hedges. Solventum may de-designate these cash flow hedge relationships in advance of the occurrence of the forecasted transaction. In addition, Solventum enters into foreign currency contracts that are not designated in hedging relationships to offset, in part, the impacts of changes in value of various non-functional currency denominated items including certain intercompany financing balances. At December 31, 2025, an instantaneous 10% change in applicable foreign currency spot exchange rates would have increased/decreased the aggregate fair value carrying amount of foreign exchange forward and cross currency swaps by approximately $42 million and $135 million, respectively.
Interest Rates Risk: The Company may be impacted by interest rate volatility with respect to existing debt and future debt issuances, and cash. Solventum manages interest rate risk and expense using a mix of fixed and floating rate debt. In addition, the Company may enter into interest rate swaps that are designated and qualify as fair value hedges. The Company's interest rate sensitivity analysis includes the impact of interest rate changes on its floating-rate notes, interest rate swap agreements, and cash balances. At December 31, 2025, an instantaneous 100 basis point change in applicable interest rates would increase/decrease the Company's pre-tax earnings by approximately $7 million on an annualized basis as it relates to the Company’s floating-rate notes, interest rate swap agreements, and interest earned on cash.
Item 8. Financial Statements and Supplementary Data
Report of Independent Registered Public Accounting Firm (PCAOB ID 238 )
Consolidated Financial Statements
Index to Financial Statements
A complete summary of Form 10-K content, including the index to financial statements, is found at the beginning of this document.
55
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Shareholders of Solventum Corporation
Opinions on the Financial Statements and Internal Control over Financial Reporting
We have audited the accompanying consolidated balance sheets of Solventum Corporation and its subsidiaries (the “Company”) as of December 31, 2025 and 2024, and the related consolidated statements of income, of comprehensive income, of changes in equity, and of cash flows for each of the three years in the period ended December 31, 2025, including the related notes (collectively referred to as the “consolidated financial statements”). We also have audited the Company's internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2025 in conformity with accounting principles generally accepted in the United States of America. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control - Integrated Framework (2013) issued by the COSO.
Basis for Opinions
The Company's management is responsible for these consolidated financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management’s Report on Internal Control over Financial Reporting. Our responsibility is to express opinions on the Company’s consolidated financial statements and on the Company's internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the consolidated financial statements included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
As described in Management’s Report on Internal Control Over Financial Reporting, management has excluded Acera Surgical (“Acera”) from its assessment of internal control over financial reporting as of December 31, 2025 because it was acquired by the Company in a purchase business combination during 2025. We have also excluded Acera from our audit of internal control over financial reporting. Acera is a wholly-owned subsidiary whose total assets and total revenues excluded from management’s assessment and our audit of internal control over financial reporting represent less than 1% of the related consolidated financial statement amounts as of and for the year ended December 31, 2025.
Definition and Limitations of Internal Control over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the
56
company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that (i) relate to accounts or disclosures that are material to the consolidated financial statements and (ii) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Revenue Recognition
As described in Note 1 to the consolidated financial statements, the majority of the Company’s customer arrangements contain a single performance obligation. For these customer arrangements, control transfers to customers at a point-in-time when goods have been delivered as that is generally when legal title, physical possession and risks and rewards of ownership transfer to the customer. The Company also enters into customer arrangements that involve multiple performance obligations (such as rental of equipment and related consumables), software with coterminous post-contract support, and software-as-a-service. For certain arrangements, specifically software sold with coterminous post-contract support that is integral to maintaining the utility of the software license to the customer and software-as-a-service, control transfers over time as the customer simultaneously receives and consumes the benefits as the Company completes the performance obligation(s). The Company recognizes rental revenue based on the length of time a device is used by the patient/organization. Revenue is recognized at the transaction price which the Company expects to be entitled. For contracts with multiple performance obligations, the Company allocates the contract’s transaction price to each performance obligation using management’s best estimate of the standalone selling price of each distinct good or service in the contract. The Company’s net sales of product and net sales of software and rentals were $6,349 million and $1,976 million, respectively, for the year ended December 31, 2025.
The principal consideration for our determination that performing procedures relating to revenue recognition is a critical audit matter is a high degree of auditor effort in performing procedures related to the Company’s revenue recognition.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the revenue recognition process. These procedures also included, among others (i) testing revenue recognized for a sample of revenue transactions from sales of product by obtaining and inspecting source documents, such as contracts, purchase orders, invoices, proof of shipment or delivery, and cash receipts; (ii) testing revenue recognized for a sample of revenue transactions from sales of software with coterminous post-contract support and software-as-a-service (collectively, “software”) by obtaining and inspecting source documents, such as contracts, purchase orders, invoices, proof of delivery, and cash receipts; (iii) testing revenue recognized for a sample of revenue transactions from arrangements involving rental equipment and related consumables (“rental”) by obtaining and inspecting source documents, such as contracts, purchase orders, invoices, proof of delivery, and cash receipts; (iv) testing a sample of sales credit transactions for product, software, and rental revenue by obtaining and inspecting source documents, such as support for the nature of the credit, including valid business purpose and application, and amount; and (v) confirming a sample of outstanding customer invoice balances as of December 31, 2025 for product, software, and rental revenue transactions and, for confirmations not returned, obtaining and inspecting source documents, such as contracts, purchase orders, invoices, proof of shipment or delivery, and subsequent cash receipts.
Divestiture of Purification and Filtration Business – Gain on Sale
As described in Notes 1, 3, and 8 to the consolidated financial statements, on February 25, 2025, the Company entered into a Transaction Agreement to sell its Purification and Filtration business to Thermo Fisher Scientific Inc. ("Buyer"). On June 25, 2025, the Company and Buyer entered into an Amended and Restated Transaction Agreement (the "Agreement") to exclude the Company’s drinking water filtration business (the "Water Business") from the scope of the Purification and Filtration business to be acquired by Buyer (such acquired business, the "Business"). On September 1, 2025, the sale of the Business to the Buyer was completed. The cash consideration paid to the Company at closing was approximately $4 billion resulting in a pre-tax gain of approximately $1.5 billion. Under the Agreement, Buyer is entitled to receive a payment of up to $75 million from the Company either upon a sale of the Water Business or after an agreed upon 3-year period. The Company recorded $64 million related to the Water Business liability within the other liabilities on the consolidated balance sheet, which was the present value of the expected future obligation at close of the Transaction. In connection with the sale of the Business, the Company entered
57
into various transition service agreements to provide certain support services at cost, below fair market value, for a period of up to 24 months from the closing of the sale. In connection with these agreements, the Company recognized an unfavorable contract liability of $113 million, which will be recognized over the terms of the transition service agreements. The Company recognized corresponding income tax impacts in various jurisdictions as a result of the gain on sale. Judgment is required in determining the Company’s tax expense and in the application of relevant tax laws and regulations to its tax positions.
The principal considerations for our determination that performing procedures relating to the gain on sale relating to the divestiture of the Purification and Filtration business is a critical audit matter are (i) the significant judgment by management when determining the gain on sale and corresponding tax effects; (ii) the high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating audit evidence related to (a) the consideration received pursuant to the Agreement, (b) the measurement of the disposal group, and (c) the application of tax laws and regulations in connection with management’s determination of the income tax effects of the divestiture; and (iii) the audit effort involved the use of professionals with specialized skill and knowledge.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the divestiture transaction and determination of corresponding tax effects. These procedures also included, among others (i) reading the Agreement; (ii) evaluating the reasonableness of management's accounting conclusions with respect to the gain on sale, including recording of the Water Business liability and unfavorable contract liabilities; (iii) testing the cash proceeds and transaction fees recorded by management; (iv) testing the completeness and accuracy of the carrying value of the disposal group and unfavorable contract liabilities; and (v) evaluating the information, including tax law and other relevant evidence, used by management to support its position regarding the tax consequences of the transaction. Professionals with specialized skill and knowledge were used to assist in the evaluation of the application of relevant tax laws and regulations.
Acquisition of Acera Surgical – Valuation of Developed Technology
As described in Note 3 to the consolidated financial statements, on December 23, 2025, the Company acquired Acera Surgical (“Acera”) for total purchase consideration of $776 million, which included cash consideration of $696 million, net of cash acquired, and a future milestone payment of an additional $125 million that had a fair value of $80 million on the acquisition date. The acquired intangible assets of $440 million consisted primarily of developed technology. The fair value of the developed technology was determined using a multi-period excess earnings method which includes significant assumptions related to revenue growth rates, EBITDA margins, obsolescence factors, and discount rate.
The principal considerations for our determination that performing procedures relating to the valuation
of developed technology acquired in the acquisition of Acera is a critical audit matter are (i) the significant judgment by management when developing the fair value estimate of the developed technology acquired; (ii) a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating management’s significant assumptions related to revenue growth rates, EBITDA margins, discount rate, and obsolescence factors for developed technology; and (iii) the audit effort involved the use of professionals with specialized skill and knowledge.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the acquisition accounting, including controls over management’s valuation of the developed technology acquired. These procedures also included, among others (i) reading the purchase agreement; (ii) testing management’s process for developing the fair value estimate of the developed technology acquired; (iii) evaluating the appropriateness of the multi-period excess earnings method used by management; and (iv) evaluating the reasonableness of the significant assumptions used by management related to revenue growth rates, EBITDA margins, obsolescence factors, and discount rate for developed technology. Evaluating management’s assumptions related to revenue growth rates and EBITDA margins for developed technology involved considering (i) the current and past performance of the Acera business; (ii) the consistency with external market and industry data; and (iii) whether the assumptions were consistent with evidence obtained in other areas of the audit. Professionals with specialized skill and knowledge were used to assist in evaluating (i) the appropriateness of the multi-period excess earnings method and (ii) the reasonableness of the discount rate and obsolescence factors assumptions for developed technology.
/s/ PricewaterhouseCoopers LLP
February 27, 2026
We have served as the Company's auditor since 2022.
58
Solventum Corporation
Consolidated Statements of Income
| Year ended December 31, | ||||||||||||||||||||
| (Millions, except per share data) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net sales of product | $ | $ | $ | |||||||||||||||||
| Net sales of software and rentals | ||||||||||||||||||||
| Total net sales | ||||||||||||||||||||
| Cost of product | ||||||||||||||||||||
| Cost of software and rentals | ||||||||||||||||||||
| Gross profit | ||||||||||||||||||||
| Selling, general and administrative expenses | ||||||||||||||||||||
| Research and development expenses | ||||||||||||||||||||
| Gain on sale of business | ( | ( | ||||||||||||||||||
| Operating income | ||||||||||||||||||||
| Interest expense, net | ||||||||||||||||||||
| Loss on debt extinguishment, net | ||||||||||||||||||||
| Other expense (income), net | ||||||||||||||||||||
| Income before income taxes | ||||||||||||||||||||
| Provision for income taxes | ||||||||||||||||||||
| Net income | $ | $ | $ | |||||||||||||||||
| Earnings per share: | ||||||||||||||||||||
| Basic earnings per share | $ | $ | $ | |||||||||||||||||
| Diluted earnings per share | ||||||||||||||||||||
| Weighted-average number of shares outstanding: | ||||||||||||||||||||
| Basic | ||||||||||||||||||||
| Diluted | ||||||||||||||||||||
The accompanying Notes to the Consolidated Financial Statements are an integral part of these statements.
59
Solventum Corporation
Consolidated Statements of Comprehensive Income
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net income | $ | $ | $ | |||||||||||||||||
| Other comprehensive income (loss), net of tax: | ||||||||||||||||||||
| Cumulative translation adjustment | ( | |||||||||||||||||||
| Defined benefit pension and postretirement plans | ( | ( | ||||||||||||||||||
| Cash flow hedging instruments | ( | |||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | |||||||||||||||||||
| Comprehensive income | $ | $ | $ | |||||||||||||||||
The accompanying Notes to the Consolidated Financial Statements are an integral part of these statements.
60
Solventum Corporation
Consolidated Balance Sheets
| December 31, | ||||||||||||||
| (Millions, except share information) | 2025 | 2024 | ||||||||||||
| Assets | ||||||||||||||
| Current assets | ||||||||||||||
| Cash and cash equivalents | $ | $ | ||||||||||||
Accounts receivable — net of allowances of $ | ||||||||||||||
| Due from related parties | ||||||||||||||
| Inventories | ||||||||||||||
| Finished goods | ||||||||||||||
| Work in process | ||||||||||||||
| Raw materials and supplies | ||||||||||||||
| Total inventories | ||||||||||||||
| Other current assets | ||||||||||||||
| Total current assets | ||||||||||||||
| Property, plant and equipment — net | ||||||||||||||
| Goodwill | ||||||||||||||
| Intangible assets — net | ||||||||||||||
| Other assets | ||||||||||||||
| Total assets | $ | $ | ||||||||||||
| Liabilities | ||||||||||||||
| Current liabilities | ||||||||||||||
| Short-term borrowings and current portion of long-term debt | $ | $ | ||||||||||||
| Accounts payable | ||||||||||||||
| Due to related parties | ||||||||||||||
| Unearned revenue | ||||||||||||||
| Other current liabilities | ||||||||||||||
| Total current liabilities | ||||||||||||||
| Long-term debt | ||||||||||||||
| Pension and postretirement benefits | ||||||||||||||
| Deferred income taxes | ||||||||||||||
| Other liabilities | ||||||||||||||
| Total liabilities | $ | $ | ||||||||||||
| Commitments and contingencies (Note 12) | ||||||||||||||
| Equity | ||||||||||||||
Common stock, par value $ | $ | $ | ||||||||||||
Shares issued and outstanding - December 31, 2025: | ||||||||||||||
Shares issued and outstanding - December 31, 2024: | ||||||||||||||
| Additional paid-in-capital | ||||||||||||||
| Retained earnings | ||||||||||||||
Accumulated other comprehensive income (loss) | ( | ( | ||||||||||||
| Total equity | ||||||||||||||
| Total liabilities and equity | $ | $ | ||||||||||||
The accompanying Notes to the Consolidated Financial Statements are an integral part of these statements.
61
Solventum Corporation
Consolidated Statements of Changes in Equity
| Common Stock | ||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | Shares Outstanding | Par Value | Additional Paid-In-Capital | Retained Earnings | Net Parent Investment | Accumulated Other Comprehensive Income (Loss) | Total | |||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2022 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Net transfers to 3M | — | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2023 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||
| Net transfers to 3M | — | — | — | — | ( | ( | ( | |||||||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
Common stock for tax withholding obligations | — | — | ( | — | — | — | ( | |||||||||||||||||||||||||||||||||||||
Issuance of common stock in connection with Spin-Off and reclassification of net parent investment | — | ( | — | |||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2024 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
Net transfers to 3M | — | — | ( | — | — | — | ( | |||||||||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||
Common stock for tax withholding obligations | — | — | ( | — | — | — | ( | |||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2025 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
The accompanying Notes to the Consolidated Financial Statements are an integral part of these statements.
62
Solventum Corporation
Consolidated Statements of Cash Flows
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Cash Flows from Operating Activities | ||||||||||||||||||||
| Net income | $ | $ | $ | |||||||||||||||||
| Adjustments to reconcile net income to net cash provided by operating activities | ||||||||||||||||||||
| Depreciation and amortization | ||||||||||||||||||||
Pension and postretirement benefit expense | ||||||||||||||||||||
| Stock-based compensation expense | ||||||||||||||||||||
| Gain on sale of business | ( | ( | ||||||||||||||||||
| Transaction costs | ( | |||||||||||||||||||
| Deferred income taxes | ( | ( | ( | |||||||||||||||||
| Changes in assets and liabilities | ||||||||||||||||||||
| Accounts receivable | ( | |||||||||||||||||||
| Due from related parties | ||||||||||||||||||||
| Inventories | ( | ( | ||||||||||||||||||
| Accounts payable | ||||||||||||||||||||
Due to related parties | ( | ( | ||||||||||||||||||
| Accrued compensation | ||||||||||||||||||||
| All other operating activities - net | ( | ( | ||||||||||||||||||
| Net cash provided by operating activities | ||||||||||||||||||||
| Cash Flows from Investing Activities | ||||||||||||||||||||
| Purchases of property, plant and equipment | ( | ( | ( | |||||||||||||||||
| Acquisitions, net of cash acquired | ( | |||||||||||||||||||
| Proceeds from sale of business | ||||||||||||||||||||
| Other — net | ( | |||||||||||||||||||
| Net cash provided by (used in) investing activities | ( | ( | ||||||||||||||||||
| Cash Flows from Financing Activities | ||||||||||||||||||||
| Repayment of debt | ( | ( | ||||||||||||||||||
| Net transfers to 3M | ( | ( | ( | |||||||||||||||||
| Proceeds from long-term debt, net of issuance costs | ||||||||||||||||||||
| Other — net | ||||||||||||||||||||
Net cash used in financing activities | ( | ( | ( | |||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | ||||||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | ||||||||||||||||||||
| Cash and cash equivalents at beginning of year | ||||||||||||||||||||
| Cash and cash equivalents at end of year | $ | $ | $ | |||||||||||||||||
| Supplemental Cash Flow Information | ||||||||||||||||||||
| Cash paid for: | ||||||||||||||||||||
| Income taxes | $ | $ | $ | |||||||||||||||||
| Interest | ||||||||||||||||||||
63
Solventum Corporation
Notes to the Consolidated Financial Statements
NOTE 1. Significant Accounting Policies
Organization and Description of Business
Solventum Corporation ("Solventum," "we," "our," "us," or the "Company") was a business of 3M Company ("3M"). On April 1, 2024 (the "Distribution Date"), 3M completed the previously announced spin-off of Solventum Corporation (the "Spin-Off"). The Spin-Off was completed through a distribution of approximately 80.1 % of the Company’s outstanding common stock to holders of record of 3M’s common stock as of the close of business on March 18, 2024 (the "Distribution"), which resulted in the issuance of 172,709,505 shares of common stock. As a result of the Distribution, the Company became an independent public company. Solventum’s common stock is listed under the symbol "SOLV" on the New York Stock Exchange ("NYSE").
Solventum is a leading global healthcare company with a broad portfolio of trusted solutions that leverage deep material science, data science, and digital capabilities to address critical customer needs. Solventum is organized into three
Basis of Presentation
The consolidated financial statements have been prepared in accordance with U.S. generally accepted accounting principles ("GAAP") and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission ("SEC") and present the historical results of operations, comprehensive income, and cash flows for the years ended December 31, 2025, 2024, and 2023, and financial position as of December 31, 2025 and 2024.
The Company’s financial statements are presented on a consolidated basis. Prior to April 1, 2024, Solventum was a carve-out business of 3M. The Company's financial statements prior to April 1, 2024 were prepared on a combined basis and were derived from the consolidated financial statements and accounting records of 3M, including the historical cost basis of assets and liabilities comprising the Company, as well as the historical revenues, direct costs, and allocations of indirect costs attributable to the operations of the Company, using the historical accounting policies applied by 3M. The financial statements included in this annual report are referred to as the "Consolidated Financial Statements" for all periods presented.
All amounts discussed are in millions of U.S. dollars, unless otherwise indicated. Amounts reported within this annual report are rounded to the nearest million and the sum of the components may not equal the total amount reported due to rounding. Additionally, certain columns and rows within tables may not sum due to rounding.
Foreign currency translation: Local currencies generally are considered the functional currencies outside the United States, and accordingly, the financial statements of these subsidiaries are remeasured as if their functional currency is that of their parent. Assets and liabilities for operations in local-currency environments are translated at month-end exchange rates of the period reported. Income and expense items are translated at average monthly currency exchange rates in effect during the period. Cumulative translation adjustments are recorded as a component of accumulated other comprehensive income (loss) in the consolidated balance sheets.
Use of estimates: The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. Such estimates and assumptions are subject to inherent uncertainties which may result in actual amounts differing from those estimates.
Cash and cash equivalents: Cash and cash equivalents included in the Company's consolidated balance sheets includes cash deposits, certificates of deposit, money market and other short-term funds with original maturities of three months or less.
64
Inventories: Inventories are stated at the lower of cost or net realizable value (NRV), which is defined as estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal and transportation. Cost is generally determined on a first-in, first-out basis.
Business Combinations: The Company accounts for acquired businesses using the acquisition method of accounting in accordance with U.S. GAAP, which requires the assets and liabilities of the acquired businesses to be recorded and consolidated on the acquisition date at their respective fair values. The Company's business combinations typically result in the recognition of goodwill, developed technology, and other intangible assets. The results of operations from acquisitions are included in the Company's consolidated financial statements from their respective acquisition dates. Transactions costs associated with acquisitions are expensed as incurred and recorded within selling, general, and administrative expenses.
For arrangements that involve potential future contingent consideration, we record a liability at the acquisition date equal to the fair value of the consideration that we expect to pay in the future. Subsequent changes in the fair value of those contingent consideration liabilities are recognized in earnings until the underlying contingencies are resolved.
Intangible assets: Intangible asset types include customer-related, patents and technology, and tradenames and other intangible assets acquired from an independent party. Intangible assets with a definite life are amortized on a systematic and rational basis (generally straight line) that is representative of the asset’s use. The estimated useful lives vary by category, with customer-related between to nineteen years , patents and technology between to ten years , and definite lived tradenames and other between and sixteen years . Intangible assets are removed from their respective gross asset and accumulated amortization accounts when they are no longer in use. Refer to Note 4 for additional details on the gross amount and accumulated amortization of the Company’s intangible assets.
Intangible assets with a definite life are tested for impairment whenever events or circumstances indicate that the carrying amount of an asset or asset group may not be recoverable. An impairment loss is recognized when the carrying amount exceeds the estimated undiscounted cash flows from the asset’s or asset group’s ongoing use and eventual disposition. If an impairment is identified, the amount of the impairment loss recorded is calculated by the excess of the asset’s carrying value over its fair value. Fair value is generally determined using a discounted cash flow analysis.
Restructuring actions: We record liabilities for costs associated with exit activities in the period in which the liability is incurred. Employee termination costs are accrued when the restructuring is probable and estimable, which typically is when management approves the associated actions. Costs for one-time termination benefits in which the employee is required to
65
Revenue recognition: The Company sells a wide range of products to a diversified base of customers around the world. The majority of Solventum’s customer arrangements contain a single performance obligation. The Company also enters into customer arrangements that involve multiple performance obligations (such as rental of equipment and related consumables), software with coterminous post-contract support, and software-as-a-service.
The Company recognizes revenue in accordance with Accounting Standards Codification (ASC) 606, Revenue from Contracts with Customers. Revenue is recognized when control of goods has transferred to customers. For the majority of the Company’s customer arrangements, control transfers to customers at a point-in-time when goods have been delivered as that is generally when legal title, physical possession and the risks and rewards of ownership transfer to the customer. For certain arrangements, specifically software sold with coterminous post-contract support that is integral to maintaining the utility of the software license to the customer and software-as-a-service, control transfers over time as the customer simultaneously receives and consumes the benefits as the Company completes the performance obligation(s).
Revenue is recognized at the transaction price which the Company expects to be entitled. The transaction price includes customer rebates, trade promotion funds, and cash discounts, which are accounted for as variable consideration. These sales incentives are recorded as a reduction to revenue at the time of the initial sale based on the most-likely estimate. The most-likely estimate considers historical experience, trend analysis, and projected market conditions in the various markets served. Because the Company serves numerous markets, the sales incentive programs offered vary across businesses, but the most common incentive relates to amounts paid or credited to customers for achieving defined volume levels or growth objectives. There are no material instances where variable consideration is constrained and not recorded at the initial time of sale. Sales, use, value-added, and other excise taxes are not recognized in revenue. The Company has elected to present revenue net of sales taxes and other similar taxes.
Product returns are recorded as a reduction to revenue based on anticipated sales returns that occur in the normal course of business. The Company primarily has assurance-type warranties that do not result in separate performance obligations.
For contracts with multiple performance obligations, the Company allocates the contract’s transaction price to each performance obligation using the Company’s best estimate of the standalone selling price of each distinct good or service in the contract. For customers purchasing software from the Company, these performance obligations include providing software licenses with ongoing customer support, installation and training. For medical equipment arrangements, performance obligations include both furnishing a rental unit to a customer as well as delivery of consumables.
The Company recognizes revenue from the rental of equipment in accordance with the guidance of ASC 842, Leases. The Company recognizes rental revenue based on the length of time a device is used by the patient/organization, (i) at the contracted rental rate for contracted customers, and (ii) generally, retail price for non-contracted customers. The leases are short-term in nature and are all classified as operating leases.
The Company does not have material unfulfilled performance obligation balances for contracts with an original length greater than one year in any years presented. Additionally, the Company does not have material costs related to obtaining a contract with amortization periods greater than one year for any year presented.
Accounts receivable and allowances: Trade accounts receivable are recorded at the invoiced amount and do not bear interest. The Company maintains allowances for bad debts, which are based on the best estimate of the amount of expected credit losses in existing accounts receivable. The Company determines the allowances based on historical write-off experience informed by industry and regional economic data, and current expectations of future credit losses. The Company reviews the allowances monthly. The allowances for bad debts as well as the provision for credit losses, write-off activity and recoveries for the periods presented are not material.
Software costs: The Company capitalizes direct costs associated with the development of, and external software acquired for use as, internal-use software. Capitalized software costs are included in property, plant and equipment on the consolidated balance sheets. Amounts capitalized are amortized over a period of to seven years , generally on a straight-line basis, unless another systematic and rational basis is more representative of the software’s use.
66
Income taxes: Prior to the Spin-Off, the Company was included in the consolidated U.S. federal, state, and foreign income tax returns of 3M, where applicable, through April 1, 2024. The Company's income tax provision for the periods 2023 and the first quarter of 2024 was prepared using the separate return method. The calculation of income taxes on a separate return basis requires a considerable amount of judgment and use of both estimates and allocations. As a result, transactions included in the consolidated financial statements of 3M may not be included in the Company's consolidated financial statements. Similarly, the tax treatment of certain items reflected in the Company's consolidated financial statements may not be reflected in the consolidated financial statements and tax returns of 3M. Therefore, items such as net operating losses, credit carryforwards, and valuation allowances may exist in the Company's standalone financial statements that may or may not exist in 3M’s consolidated financial statements. After the Spin-Off, the Company has filed tax returns on its own behalf, and its income tax provision was prepared on a standalone basis. As a result, its deferred taxes and effective tax rate for 2025 may differ from those in the historical periods prior to Spin-Off.
In jurisdictions where the Company has been included in income tax returns filed by 3M, income taxes currently payable will be deemed to have been remitted to 3M, in cash, in the period the liability arose and income taxes currently receivable are deemed to have been received from the Parent in the period that a refund could have been recognized. Adjustments to the recorded payable that derive from the Company's current year activity are recorded through current tax expenses and the ending adjusted payable/receivable is settled through "Net parent investment" on the consolidated balance sheets.
Current obligations for tax in jurisdictions where the Company does not file a consolidated tax return with 3M, including certain foreign and certain U.S. state tax jurisdictions, are recorded as accrued liabilities within "Other current liabilities" on the consolidated balance sheets. The effects of tax adjustments and settlements with taxing authorities are presented in our consolidated financial statements in the period to which they relate.
Judgment is required in determining the Company’s tax expense and in the application of relevant tax laws and regulations to its tax positions. Uncertain tax positions that meet the more likely than not recognition threshold are measured to determine the amount of tax benefit to recognize in the consolidated financial statements. An uncertain tax position is measured at the largest amount of benefit that the Company believes has a greater than 50 percent likelihood of realization upon settlement. Tax benefits not meeting the measurement or realization criteria represent unrecognized tax benefits. The Company recognizes interest and penalties related to income tax matters as a component of "Provision for income taxes" in the consolidated statements of income.
Deferred income tax balances reflect the effects of temporary differences between the carrying amounts of assets and liabilities and their respective tax bases, as well as from net operating loss and tax credit carryforwards. The deferred income tax balances are stated at enacted tax rates expected to be in effect when those taxes are paid or recovered. Deferred income tax assets represent amounts available to reduce income taxes payable on taxable income in future years. We evaluate the recoverability of these future tax deductions and tax credits by evaluating all available positive and negative evidence, specifically assessing the adequacy of future expected taxable income from all sources, including reversal of existing taxable temporary differences, forecasted operating earnings, and available tax planning strategies. To the extent we consider it more likely than not that a deferred tax asset will not be recovered, a valuation allowance is established.
Comprehensive income: Total comprehensive income and the components of accumulated other comprehensive income (loss) are presented in the consolidated statements of comprehensive income and the consolidated statements of changes in equity. Accumulated other comprehensive income (loss) is composed of foreign currency translation effects, defined benefit pension adjustments and gains/losses from cash flow hedging activity.
67
Fair value measurements: Solventum follows ASC 820, Fair Value Measurements, with respect to assets and liabilities that are measured at fair value on a recurring basis and nonrecurring basis. Under the standard, fair value is defined as the exit price, or the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants as of the measurement date. The standard also establishes a hierarchy for inputs used in measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that the most observable inputs be used when available. Observable inputs are inputs market participants would use in valuing the asset or liability developed based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the factors market participants would use in valuing the asset or liability developed based upon the best information available in the circumstances. The hierarchy is broken down into three levels. Level 1 inputs are quoted prices (unadjusted) in active markets for identical assets or liabilities. Level 2 inputs include quoted prices for similar assets or liabilities in active markets, quoted prices for identical or similar assets or liabilities in markets that are not active, and inputs (other than quoted prices) that are observable for the asset or liability, either directly or indirectly. Level 3 inputs are unobservable inputs for the asset or liability. Categorization within the valuation hierarchy is based upon the lowest level of input that is significant to the fair value measurement. The carrying value of the Company's accounts receivable, accounts payable, and accrued expenses approximate their fair value due to the short period of time to maturity or repayment.
Leases: Solventum determines if an arrangement is a lease upon inception. A contract is or contains a lease if the contract conveys the right to control the use of an identified asset for a period of time in exchange for consideration. The right to control the use of an asset includes the right to obtain substantially all of the economic benefits of the underlying asset and the right to direct how and for what purpose the asset is used.
Operating lease right-of-use assets and liabilities are recognized at commencement date based on the present value of lease payments over the lease term. As the Company’s leases typically do not provide an implicit rate, the present value of our lease liability is determined using Solventum’s incremental borrowing rate at lease commencement. Solventum determines the incremental borrowing rate for leases using a portfolio approach based primarily on the lease term and the economic environment of the applicable country or region.
Finance lease right-of-use assets and liabilities are generally those leases for which the Company will pay substantially all the underlying asset’s fair value or will use the asset for all or a major part of its economic life, including circumstances in which the Company will ultimately own the asset. Finance lease assets are included in property, plant, and equipment - net, and finance lease liabilities are included in other current liabilities and other liabilities on the consolidated balance sheets. For finance leases, the Company recognizes interest expense using the effective interest method, and the Company recognizes amortization expense on the lease right-of-use asset over the shorter of the lease term or the useful life of the asset. When a finance lease includes a purchase option that the Company is reasonably certain to exercise, the right-of-use asset is amortized over the useful life of the asset.
As a lessee, the Company leases distribution centers, office space, land, and equipment. Certain lease agreements include rental payments adjusted annually based on changes in an inflation index. The Company’s leases do not contain material residual value guarantees or material restrictive covenants. Lease expense is recognized on a straight-line basis over the lease term.
Certain leases include one or more options to renew, with terms that can extend the lease term up to five years . The Company includes options to renew the lease as part of the right-of-use lease asset and liability when it is reasonably certain the Company will exercise the option. In addition, certain leases contain fair value purchase and termination options with an associated penalty. In general, the Company is not reasonably certain to exercise such options.
For the measurement and classification of its lease agreements, the Company groups lease and non-lease components into a single lease component for all underlying asset classes. Variable lease payments primarily include payments for non-lease components, such as maintenance costs, payments for leased assets used beyond their noncancellable lease term as adjusted for contractual options to terminate or renew, additional payments related to a subsequent adjustment in an inflation index, and payments for non-components such as sales tax. Certain leases contain immaterial variable lease payments based on number of units produced.
68
New Accounting Pronouncements
The table below provides summaries of recently adopted financial accounting standards and recently issued financial accounting standards.
| Standard | Relevant Description | Effective Date for Solventum | Impact of Adoption | ||||||||
ASU No. 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures | Issued in December 2023. Requires disaggregated information about a Company's effective tax rate reconciliation as well as information on income taxes paid. | Year-end December 31, 2025 | The Company has assessed the impact of the updated standard and has included expanded disclosures within the income taxes footnote. | ||||||||
ASU No. 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses | Issued in November 2024. Requires additional disclosure of the nature of expenses included in the income statement. | Year-end December 31, 2026 | The Company is currently assessing the impact that the updated standard will have on financial statement disclosures. | ||||||||
ASU No. 2025-06, Intangibles—Goodwill and Other—Internal-Use Software (Subtopic 350-40): Targeted Improvements to the Accounting for Internal-Use Software | Issued September 2025. Updates existing accounting guidance regarding the capitalization of internal-use software costs. | January 1, 2026 | The Company has elected to early adopt this standard on a prospective basis and the early adoption of this standard will not have a material impact on the Company's financial statement disclosures. | ||||||||
NOTE 2. Revenue Recognition
Contract Balances
Unearned revenue primarily relates to revenue that is recognized over time for one-year software license contracts. Approximately $550 million, $550 million, and $540 million of the December 31, 2024, 2023, and 2022 balance, respectively, was recognized as revenue during the years ended December 31, 2025, 2024, and 2023, respectively.
Operating Lease Revenue
includes rental revenue from durable medical devices as part of operating lease arrangements (reported within the MedSurg segment), which was $615 million, $600 million, and $616 million for the years ended December 31, 2025, 2024, and 2023, respectively.
Customer Concentration
No customer accounted for more than 10% of the Company’s revenues for the years ended December 31, 2025, 2024, or 2023. Additionally, no customers accounted for more than 10% of accounts receivable as of December 31, 2025 and 2024.
NOTE 3. Acquisitions and Divestitures
Acquisitions
The Company had an acquisition during the year ended 2025 that was accounted for as a business combination. The Company had no acquisitions in 2024.
Year Ended 2025 Acquisitions
On December 23, 2025, the Company acquired Acera Surgical ("Acera"), a privately held bioscience company focused on developing and commercializing fully engineered materials for regenerative wound care that expands the Company's acute care portfolio, pursuant to a merger agreement. For the year ended December 31, 2025, net sales related to Acera were not material to the Company's results of operations or financial condition.
Total purchase consideration was $776 million, which included cash consideration of $696 million, net of cash acquired, and a future milestone payment that had a fair value of $80
69
The future payment owed under the milestone is $125 million. The acquisition date fair value of the contingent consideration was determined using a Monte Carlo simulation, which considered significant unobservable inputs, including management's projection of net sales, revenue volatility and revenue risk premium. Revenue volatility and revenue risk premium were estimated at 45 % and 8.9 %, respectively. The fair value measurement of the contingent consideration is classified as level 3 within the fair value hierarchy. The fair value of the contingent milestone payment will be remeasured each quarter, with changes in the fair value recognized within the consolidated statements of income.
In connection with the acquisition, the Company agreed to pay $18 million in transaction expenses incurred by the acquiree, which were treated as an assumed liability at close. Acquisition-related expenses of $7 million incurred by the Company were recorded in selling, general, and administrative expenses.
The fair value of the acquisition was allocated to the assets acquired of $488 million, consisting primarily of developed technology and other amortizable intangibles of $440 million, goodwill of $441 million, and liabilities assumed of $131 million, which includes deferred taxes of $94 million. The fair value of the developed technology was determined using a multi-period excess earnings method, which includes significant assumptions related to revenue growth rates, EBITDA margins, obsolescence factors, and discount rate. The weighted-average useful life of the acquired intangible assets is approximately eight years . The goodwill is primarily related to synergies expected to be achieved from the addition of Acera's synthetic tissue matrix technology into Solventum's existing advanced wound care portfolio. The goodwill is included in the Company’s MedSurg segment and is not deductible for tax purposes.
The purchase price allocations are considered preliminary as of December 31, 2025, with final estimates of fair value expected to be completed in the first half of 2026, but no later than one year after the acquisition date. A change in the estimated fair value of the net assets acquired will result in a corresponding change to the amount of the purchase price allocated to goodwill.
Divestitures
Year Ended 2025 Divestitures
On February 25, 2025, the Company entered into a Transaction Agreement to sell its Purification and Filtration business to Thermo Fisher Scientific Inc. ("Buyer"). On June 25, 2025, the Company and Buyer entered into an Amended and Restated Transaction Agreement (the "Agreement") to exclude the Company’s drinking water filtration business (the "Water Business") from the scope of the Purification and Filtration business to be acquired by Buyer (such acquired business, the "Business").
On September 1, 2025, Solventum completed the sale of the Business to the Buyer. The cash consideration paid to Solventum at closing was approximately $4 billion, resulting in a pre-tax gain of approximately $1.5 billion. The pre-tax gain was recorded net of transaction fees of $86 million.
Under the Agreement, Buyer is entitled to receive a payment of up to $75 million from the Company either upon a sale of the Water Business or after an agreed upon 3-year period. The Company recorded $64 million within the other liabilities on the consolidated balance sheets, which was the present value of the expected future obligation at close of the Transaction.
In connection with the sale of the Business, the Company entered into various transition service agreements to provide certain support services at cost, below fair market value, for a period of up to 24 months from the closing of the sale. These transition services include product distribution and supply chain management, contract manufacturing, information technology, finance, customer support, among others. In connection with these agreements, the Company recognized an unfavorable contract liability of $113 million, which will be recognized over the terms of the agreements.
70
NOTE 4. Goodwill and Intangible Assets
Goodwill
The goodwill balance by business segment is as follows:
| (Millions) | MedSurg | Dental Solutions | Health Information Systems | Purification and Filtration | All Other | Total | ||||||||||||||||||||||||||||||||
| Balance as of December 31, 2023 | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
| Translation impact | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||
| Balance as of December 31, 2024 | ||||||||||||||||||||||||||||||||||||||
| Acquisition activity | ||||||||||||||||||||||||||||||||||||||
| Divestiture activity | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Translation and other | ( | |||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2025 | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
The Company completed its annual goodwill impairment test in the fourth quarter of 2025 for all reporting units and determined that there was no impairment.
For the year ended December 31, 2025, there was $441 million of goodwill recorded from the acquisition of Acera on December 23, 2025. There were no acquisitions completed in 2024.
Acquired Intangible Assets: The carrying amount and accumulated amortization of acquired finite-lived intangible assets are as follows:
| December 31, | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
Customer related | $ | $ | ||||||||||||
Patents and technology | ||||||||||||||
Tradenames and other | ||||||||||||||
| Total gross carrying amount | ||||||||||||||
| Accumulated amortization — customer related | ( | ( | ||||||||||||
| Accumulated amortization — patents and technology | ( | ( | ||||||||||||
| Accumulated amortization — tradenames and other | ( | ( | ||||||||||||
| Total accumulated amortization | ( | ( | ||||||||||||
| Total intangible assets — net | $ | $ | ||||||||||||
Amortization expense was as follows:
Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Amortization expense | $ | $ | $ | |||||||||||||||||
Expected amortization expense for acquired amortizable intangible assets recorded as of December 31, 2025 is as follows:
| (Millions) | 2026 | 2027 | 2028 | 2029 | 2030 | After 2030 | ||||||||||||||||||||||||||||||||
| Amortization expense | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
71
NOTE 5. Supplemental Financial Information
Other current assets included in the consolidated balance sheets consist of the following:
| December 31, | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
| Other current assets | ||||||||||||||
| Purification and Filtration-related | $ | $ | ||||||||||||
| Prepaid income taxes | ||||||||||||||
| Prepaid IT expenses | ||||||||||||||
| Other | ||||||||||||||
| Total other current assets | $ | $ | ||||||||||||
Purification and Filtration-related in the table above includes receivables and other assets associated with post-closing activity with Buyer.
Other current liabilities included in the consolidated balance sheets consist of the following:
| December 31, | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
| Other current liabilities | ||||||||||||||
| Accrued compensation | $ | $ | ||||||||||||
Accrued taxes | ||||||||||||||
Accrued rebates | ||||||||||||||
| Accrued interest | ||||||||||||||
| Purification and Filtration-related | ||||||||||||||
| Other | ||||||||||||||
| Total other current liabilities | $ | $ | ||||||||||||
Purification and Filtration-related in the table above includes the current portion of the unfavorable contract liability as described in Note 3, along with other activity with Buyer, including amounts owed under the transition agreements, since the close of the Transaction.
NOTE 6. Property, Plant, and Equipment - Net
Property, plant and equipment - net consisted of the following:
| December 31, | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
| Property, plant and equipment - at cost | ||||||||||||||
Buildings and leasehold improvements | $ | $ | ||||||||||||
Machinery and equipment | ||||||||||||||
| Construction in progress | ||||||||||||||
| Gross property, plant and equipment | ||||||||||||||
| Accumulated depreciation | ( | ( | ||||||||||||
| Property, plant and equipment - net | $ | $ | ||||||||||||
Depreciation expense consisted of the following:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Depreciation expense | $ | $ | $ | |||||||||||||||||
NOTE 7. Supplemental Equity and Comprehensive Income Information
Share Repurchase Program
In November 2025, Solventum's Board of Directors approved a share repurchase program, which authorizes the Company to purchase up to $1 billion of the Company's outstanding common stock. There were no repurchases made under this program in 2025.
72
Changes in Accumulated Other Comprehensive Income (Loss) by Component
The table below presents the changes in accumulated other comprehensive income (loss) ("AOCI"), including the reclassifications out of AOCI by component:
| (Millions) | Cumulative Translation Adjustment | Defined Benefit Pension | Cash Flow Hedging | Total Accumulated Other Comprehensive Income (Loss) | ||||||||||||||||||||||
| Balance at December 31, 2022, net of tax | $ | ( | $ | $ | $ | ( | ||||||||||||||||||||
| Other comprehensive income (loss), before tax: | ||||||||||||||||||||||||||
| Amounts before reclassifications | ( | |||||||||||||||||||||||||
| Amounts reclassified out | ||||||||||||||||||||||||||
| Total other comprehensive income (loss), before tax | ( | |||||||||||||||||||||||||
| Tax effect | ||||||||||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | |||||||||||||||||||||||||
| Transfers from 3M, net of tax | ||||||||||||||||||||||||||
| Balance at December 31, 2023, net of tax: | $ | ( | $ | $ | $ | ( | ||||||||||||||||||||
| Other comprehensive income (loss), before tax: | ||||||||||||||||||||||||||
| Amounts before reclassifications | ( | ( | ( | |||||||||||||||||||||||
| Amounts reclassified out | ||||||||||||||||||||||||||
| Total other comprehensive income (loss), before tax | ( | ( | ( | |||||||||||||||||||||||
| Tax effect | ( | ( | ( | |||||||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | ( | ( | |||||||||||||||||||||||
| Transfers from 3M, net of tax | ( | ( | ||||||||||||||||||||||||
Balance at December 31, 2024, net of tax: | $ | ( | $ | ( | $ | $ | ( | |||||||||||||||||||
| Other comprehensive income (loss), before tax: | ||||||||||||||||||||||||||
| Amounts before reclassifications | ( | ( | ||||||||||||||||||||||||
| Amounts reclassified out | ( | |||||||||||||||||||||||||
| Total other comprehensive income (loss), before tax | ( | |||||||||||||||||||||||||
| Tax effect | ( | |||||||||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | |||||||||||||||||||||||||
| Balance at December 31, 2025, net of tax | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||
Additional details on the amounts reclassified from AOCI into consolidated income include:
•Cumulative translation adjustment: amounts were reclassified into other expense (income), net and were related to charges associated with the substantial liquidation of foreign operations completed as part of our separation from 3M.
•Defined benefit pension and postretirement plans: amounts were reclassified into other expense (income), net (see Note 10).
•Cash flow hedging: foreign currency forward contracts amounts were reclassified into cost of sales (see Note 11).
•The tax effects, if applicable, associated with these reclassifications were reflected in provision for income taxes.
73
NOTE 8. Income Taxes
Income (loss) before income taxes consisted of the following:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| United States | $ | $ | ( | $ | ||||||||||||||||
| International | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
Provision (benefit) for income taxes consisted of the following:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Currently payable | ||||||||||||||||||||
| Federal | $ | $ | $ | |||||||||||||||||
| State | ||||||||||||||||||||
| International | ||||||||||||||||||||
| Deferred | ||||||||||||||||||||
| Federal | ( | ( | ||||||||||||||||||
| State | ( | ( | ( | |||||||||||||||||
| International | ( | ( | ( | |||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
Components of deferred tax assets and (liabilities) are comprised of the following:
| December 31, | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
| Deferred tax assets: | ||||||||||||||
| Miscellaneous accruals | $ | $ | ||||||||||||
| Accrued compensation | ||||||||||||||
| Net operating/capital loss carryforward | ||||||||||||||
| Disallowed interest carryforwards | ||||||||||||||
| Foreign tax credits | ||||||||||||||
| Research and experimentation capitalization | ||||||||||||||
| Lease liabilities | ||||||||||||||
| Other deferred tax assets | ||||||||||||||
| Derivatives | ( | |||||||||||||
| Gross deferred tax assets | ||||||||||||||
| Valuation allowance | ( | ( | ||||||||||||
| Total deferred tax assets | ||||||||||||||
| Deferred tax liabilities: | ||||||||||||||
| Property, plant, and equipment | ( | ( | ||||||||||||
| Intangible assets | ( | ( | ||||||||||||
| Right-of-use assets | ( | ( | ||||||||||||
| Total deferred tax liabilities | ( | ( | ||||||||||||
| Net deferred tax asset (liability) | $ | $ | ||||||||||||
74
As displayed in the table above, as of December 31, 2025, the Company has provided $37 million of valuation allowance against certain of these deferred tax assets based on management’s determination that it is more-likely-than-not that the tax benefits related to these assets will not be realized.
As of December 31, 2025, the Company had tax-effected operating loss and capital loss carryforwards of $67 million and tax credit carryforwards of $43 million for federal, state, and international jurisdictions, with all amounts before limitation impacts and valuation allowances. Federal and international tax attributes will expire after one to an indefinite carryover period and state tax attributes will expire after one to eighteen years.
As a result of our prospective adoption of ASU 2023-09, the following table presents income taxes paid, net of refunds, disaggregated by jurisdiction in accordance with the new disclosure requirements is provided below:
| Year ended December 31, | ||||||||
| (Millions) | 2025 | |||||||
| U.S. federal | $ | |||||||
| U.S. state and local | ||||||||
| International | ||||||||
| China | ||||||||
| Germany | ||||||||
| Ireland | ||||||||
| Other international | ||||||||
| Total | $ | |||||||
As previously disclosed, cash payments directly to tax authorities were $244 million for the year ended December 31, 2024.
75
In accordance with our prospective adoption of ASU 2023-09, the following table presents a reconciliation of the U.S. federal statutory income tax rate to Solventum's worldwide effective income tax rate is provided below:
| Year ended December 31, | ||||||||||||||
| 2025 | ||||||||||||||
| (Millions) | Amount | Percent | ||||||||||||
| U.S. federal statutory income tax rate | $ | % | ||||||||||||
State and local income taxes, net of federal income tax effect (a) | ||||||||||||||
| Foreign tax effects | ||||||||||||||
| Cayman Islands | ||||||||||||||
| Statutory tax rate difference between Cayman Islands and United States | ( | ( | ||||||||||||
| Ireland | ||||||||||||||
| Statutory tax rate difference between Ireland and United States | ( | ( | ||||||||||||
| Other | ||||||||||||||
| Germany | ||||||||||||||
| Statutory tax rate difference between Germany and United States | ||||||||||||||
| Trade tax | ( | ( | ||||||||||||
| Other | ||||||||||||||
| Japan | ||||||||||||||
| Purification and Filtration Divestiture | ||||||||||||||
| Other | ( | ( | ||||||||||||
| Other foreign jurisdictions | ||||||||||||||
| Effect of changes in tax laws or rates exacted in the current period | ||||||||||||||
| Effect of cross-border tax laws | ||||||||||||||
| Global Intangible Low Taxed Income (GILTI) | ||||||||||||||
| Other | ||||||||||||||
| Tax credits | ( | ( | ||||||||||||
| Changes in valuation allowances | ||||||||||||||
| Nontaxable or nondeductible items | ||||||||||||||
| Changes in unrecognized tax benefit | ( | ( | ||||||||||||
| Other adjustments | ||||||||||||||
| Purification and Filtration Divestiture | ( | ( | ||||||||||||
| Other | ( | ( | ||||||||||||
| Effective tax rate | $ | % | ||||||||||||
(a)State and local taxes in Illinois, Pennsylvania, and Maryland made up the majority (greater than 50 percent) of the tax effect in this category.
76
A reconciliation of the U.S. federal statutory income tax rate to Solventum's worldwide effective income tax rate is provided below:
| Year ended December 31, | ||||||||||||||
| (Millions) | 2024 | 2023 | ||||||||||||
| U.S. Statutory income tax rate | % | % | ||||||||||||
| State income taxes - net of federal benefit | ||||||||||||||
| International income taxes - net | ( | ( | ||||||||||||
| Global Intangible Low Taxed Income (GILTI) | ||||||||||||||
| Foreign Derived Intangible Income (FDII) | ( | ( | ||||||||||||
| U.S. research and development credit | ( | ( | ||||||||||||
| Reserves for tax contingencies | ( | ( | ||||||||||||
| Tax impact of legal entity restructuring | ||||||||||||||
| Changes in valuation allowance | ||||||||||||||
| Deferred rate change | ||||||||||||||
| All other - net | ||||||||||||||
| Effective income tax rate | % | % | ||||||||||||
The effective income tax rate for 2025 was 9.2 %, compared to 20.9 % in 2024, a decrease of 11.7 percentage points. The primary factors that decreased the tax rate were the tax impacts of the Purification and Filtration divestiture recognized in various jurisdictions and favorable changes in the jurisdictional mix of earnings.
The Company recognizes the amount of income tax benefit that has a greater than 50% likelihood of being ultimately realized upon settlement. Changes in unrecognized tax benefits impacting the provision for income taxes of the Company have been reflected in the consolidated statements of income. Interest and penalties are also recognized in the provision for income taxes in the consolidated statements of income. For uncertain tax positions that the Company expects to be legally liable for, the unrecognized tax benefits and interest and penalties of $49 million and $36 million have been recorded to "Other liabilities" as of December 31, 2025 and 2024, respectively on the consolidated balance sheets. For uncertain tax positions where the Company is not legally and directly liable for, unrecognized tax benefits and interest and penalties are charged to "Net parent investment" on the consolidated balance sheets. A rollforward of unrecognized tax benefits ("UTB") is as follows:
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Gross UTB Balance at January 1 | $ | $ | $ | |||||||||||||||||
| Increase (decrease) for tax positions of the current year | ( | |||||||||||||||||||
| Increase (decrease) for tax positions of prior years | ( | ( | ( | |||||||||||||||||
| Settlements with tax authorities | ||||||||||||||||||||
| Reductions due to lapse of applicable statute of limitations | ( | ( | ( | |||||||||||||||||
| Reductions for amounts recorded to net parent investment | ( | |||||||||||||||||||
| Gross UTB Balance at December 31 | $ | $ | $ | |||||||||||||||||
The total amount of UTB, if recognized, would affect the effective tax rate by $127 million as of December 31, 2025. The Company has ongoing federal, state and international income tax audits in various jurisdictions and evaluates uncertain tax positions that may be challenged by local tax authorities and not fully sustained. These uncertain tax positions are reviewed on an ongoing basis and adjusted in light of facts and circumstances including progression of tax audits, developments in case law and closing statutes of limitation.
There is audit activity in several U.S. Federal, state and foreign jurisdictions where the Company is subject to ongoing tax examinations and governmental assessments, which could be impacted by evolving political environments in those jurisdictions. As of December 31, 2025, no taxing authority proposed significant adjustments to the Company’s tax positions for which the Company is not adequately reserved.
77
The Company recognizes interest and penalties accrued related to unrecognized tax benefits in tax expense. The Company recognized in the consolidated statements of income on a gross basis approximately $0.1 million of benefit, $1.0 million of benefit, and $7.5 million of benefit in 2025, 2024 and 2023, respectively. The amount of interest and penalties recognized may be an expense or benefit due to new or remeasured unrecognized tax benefit accruals. At December 31, 2025 and 2024, accrued interest and penalties in the consolidated balance sheets on a gross basis were $5.3 million and $5.3 million, respectively.
As of December 31, 2025, the Company provides for deferred taxes associated with foreign earnings in certain subsidiaries that are not considered permanently reinvested. The Company has not provided deferred taxes on approximately $1 billion of undistributed earnings from non-U.S. subsidiaries which are indefinitely reinvested in operations. It is not practical to determine the income tax liability that would be payable if such earnings were not reinvested indefinitely.
NOTE 9. Long-Term Debt and Short-Term Borrowings
Carrying value includes the impact of debt issuance costs. Long-term debt and short-term borrowings as of December 31, 2025 and 2024 consisted of the following:
| (Millions) | Currency/ Fixed vs. Floating | Effective Interest Rate | Final Maturity Date | Carrying Value | ||||||||||||||||||||||||||||
| Description | December 31, 2025 | December 31, 2024 | ||||||||||||||||||||||||||||||
month senior term loan credit facility | USD Floating | % | 2025 | $ | $ | |||||||||||||||||||||||||||
year senior term loan credit facility | USD Floating | 2027 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2027 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2029 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2031 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2034 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2054 | ||||||||||||||||||||||||||||||
$ | USD Fixed | 2064 | ||||||||||||||||||||||||||||||
| Other borrowings | ||||||||||||||||||||||||||||||||
| Total long-term debt | ||||||||||||||||||||||||||||||||
| Less: current portion of long-term debt | ||||||||||||||||||||||||||||||||
| Long-term debt (excluding current portion) | $ | $ | ||||||||||||||||||||||||||||||
Senior Notes
The Company’s borrowings include $5.0 billion aggregate principal amount of senior notes with maturity dates ranging from 2027 through 2064 (collectively, the "Senior Notes"). The Senior Notes are governed by an indenture and supplemental indenture between the Company and a trustee (collectively, the "Indenture"). The Indenture contains certain customary affirmative and negative covenants, including restrictions on the Company’s ability to consolidate, merge, convey, transfer or lease substantially all of its assets. In addition, the Indenture contains other customary terms, including certain events of default, upon the occurrence of which the Senior Notes may be declared immediately due and payable. The Company is in compliance with all covenants related to the Senior Notes.
In September 2025, the Company repurchased multiple series of its outstanding Senior Notes via cash tender offers with a combined aggregate principal amount of $1.9 billion: $650 million aggregate principal amount of 5.45 % senior notes due 2027, $797 million aggregate principal amount of 5.40 % senior notes due 2029, $22 million aggregate principal amount of 5.90 % senior notes due 2054, and $458 million aggregate principal amount of 6.00 % senior notes due 2064. Neither the 5.45 % senior notes due 2031 nor the 5.60 % senior notes due 2034 were tendered.
As a result of tendering a portion of its Senior Notes, the Company recognized a loss on debt extinguishment of approximately $94 million within loss on debt extinguishment, net within its consolidated statements of income, the cash outflow for which is recorded within other financing activities within its consolidated statements of cash flows. This loss on extinguishment reflects the difference between the carrying value and the amount paid to acquire the tendered Senior Notes and related expenses.
Credit Facilities
On February 16, 2024, the Company entered into credit agreements providing for:
•a five year senior unsecured revolving credit facility in an aggregate committed amount of $2.0 billion expiring in 2029 (the "5 -year Revolving Credit Facility"); and
78
•an eighteen month senior unsecured term loan credit facility in an aggregate principal amount of $500 million and a three year senior unsecured term credit loan facility in an aggregate principal amount of $1.0 billion (together, the "Term Loan Credit Facilities," and together with the 5 -Year Revolving Credit Facility, the "Credit Facilities"). The Term Loan Credit Facilities have a floating interest rate based on a Secured Overnight Financing Rate ("SOFR") index.
At December 31, 2025, there are no amounts outstanding under the 5 -year Revolving Credit Facility.
There were no amounts outstanding under the eighteen month senior unsecured term loan facility when it reached maturity in August 2025. For the year ended 2025, the Company prepaid $870 million of the aggregate principal amount outstanding under the three year senior unsecured term loan credit facility.
Commercial Paper
On March 4, 2024, the Company entered into a commercial paper program that allows it to issue up to $2.0 billion aggregate principal amount of short-term notes to finance short-term liabilities. Any such issuance will mature within 364 days from date of issue. There was no commercial paper outstanding as of December 31, 2025.
Future Maturities of Long-term Debt: Maturities of long-term debt in the table below reflect the impact of repayment such that total maturities equal the contractual value of long-term debt net of amounts repaid as of December 31, 2025. The maturities of long-term debt for the periods subsequent to December 31, 2025 are as follows (in millions):
| 2026 | 2027 | 2028 | 2029 | 2030 | After 2030 | Total | ||||||||||||||||||||||||||||||||
| $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
Financial Instruments Not Measured at Fair Value
NOTE 10. Pension and Postretirement Benefit Plans
Transfer of Pension and Postretirement Benefit Plans
Historically, certain employees of Solventum participated in U.S. and non-U.S. retirement plans sponsored by 3M. In preparation for the Spin-Off, certain defined benefit plan obligations and assets relating to active Solventum employees and retirees were legally transferred from 3M Company to Solventum. Upon legal transfer, the related assets and liabilities were reflected in the consolidated balance sheets. These amounts are disclosed as "Transfers from 3M" in the following tables, and the net periodic benefit costs are included in the consolidated statements of income. Prior to the legal transfer, these plans were accounted for as multiemployer plans and a proportionate allocation of service costs associated with the Solventum employees was reflected in the consolidated statements of income. Expenses associated with employees' participation in 3M Company sponsored pension plans were $5 million and $32 million for the years ended December 31, 2024 and 2023, respectively.
As of December 31, 2024, all of the defined benefit plan obligations, all associated U.S. plan assets and the majority of the associated international plan assets related to Solventum employees had transferred from 3M to Solventum. Assets that were not yet delivered from 3M-sponsored pension plans due to regulatory purposes were recognized as receivables within Solventum's plan assets as of December 31, 2024, and were transferred to Solventum in June 2025.
In total, Solventum has more than 25 defined-benefit pension plans in 14 countries. Pension benefits associated with these plans generally are based on each participant’s years of service, compensation, and age at retirement or termination. The primary U.S. defined-benefit pension plan was closed to new participants effective January 1, 2009. Prior to the Spin-Off, 3M committed to the future freeze of U.S. defined benefit pension benefits for non-union U.S. employees, effective December 31, 2028. As a successor plan to the 3M plan, the Solventum U.S. defined-benefit pension plan will also cease accruing benefits for non-union participants after December 31, 2028.
79
In addition, Solventum offers certain postretirement health care and life insurance benefits for U.S. employees who reach a retirement age while employed by the Company and who were employed prior to January 1, 2016. The related plan obligations and associated plan assets are reflected in the following tables. Most international employees and retirees are covered by government health care programs. The cost of company-provided postretirement health care plans for international employees is not material.
Assets of the U.S. qualified defined benefit pension plan and U.S. postretirement plans are held by independent trustees. There are no plan assets in the U.S. non-qualified plan due to its nature. In certain non-U.S. jurisdictions, trust funds and deposits with insurance companies are maintained to provide pension benefits to plan participants and their beneficiaries.
The following tables include a reconciliation of the beginning and ending balances of the Company-sponsored benefit obligations and the fair value of plan assets, as well as a summary of the related amounts recognized in the consolidated balance sheets as of December 31 of the respective years:
| Qualified and Non-qualified Pension Benefits | Postretirement Benefits | |||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||||||||||||||
| Change in benefit obligation | ||||||||||||||||||||||||||||||||||||||
| Benefit obligation at beginning of year | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
| Service cost | ||||||||||||||||||||||||||||||||||||||
| Interest cost | ||||||||||||||||||||||||||||||||||||||
| Participant contributions | ||||||||||||||||||||||||||||||||||||||
| Foreign exchange rate changes | ( | |||||||||||||||||||||||||||||||||||||
| Actuarial (gain) loss | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||
| Benefit payments | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||
| Settlements, curtailments, and special termination benefits | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||
| Divestitures | ( | ( | ||||||||||||||||||||||||||||||||||||
| Transfers from 3M | ||||||||||||||||||||||||||||||||||||||
| Benefit obligation at end of year | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
| Change in plan assets | ||||||||||||||||||||||||||||||||||||||
| Fair value of plan assets at beginning of year | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
| Actual return on plan assets | ||||||||||||||||||||||||||||||||||||||
| Company contributions | ||||||||||||||||||||||||||||||||||||||
| Participant contributions | ||||||||||||||||||||||||||||||||||||||
| Foreign exchange rate changes | ( | |||||||||||||||||||||||||||||||||||||
| Benefit payments | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||
| Settlements, curtailments, and special termination benefits | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Divestitures | ( | ( | ||||||||||||||||||||||||||||||||||||
| Transfers from 3M | ||||||||||||||||||||||||||||||||||||||
| Fair value of plan assets at end of year | ||||||||||||||||||||||||||||||||||||||
| Funded status at end of year | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||
80
Amounts recognized in the Consolidated Balance Sheets as of December 31, (Millions) | Qualified and Non-qualified Pension Benefits | Postretirement Benefits | ||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | |||||||||||||||||||||||||||||||||
| Other assets | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||
| Accrued benefit cost | ||||||||||||||||||||||||||||||||||||||
| Current liabilities | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||
| Non-current liabilities | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||
| Ending balance | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||
Amounts recognized in accumulated other comprehensive (income) loss as of December 31, (Millions) | Qualified and Non-qualified Pension Benefits | Postretirement Benefits | ||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | |||||||||||||||||||||||||||||||||
| Net actuarial loss (gain) | $ | $ | $ | ( | $ | $ | $ | |||||||||||||||||||||||||||||||
| Prior service cost (credit) | ( | ( | ||||||||||||||||||||||||||||||||||||
| Ending balance | $ | $ | $ | ( | $ | $ | $ | |||||||||||||||||||||||||||||||
The balance of amounts recognized for non-U.S. plans in accumulated other comprehensive income (loss) as of December 31, 2025 and December 31, 2024 in the preceding table is presented based on the foreign currency exchange rates on that date.
The pension accumulated benefit obligation represents the actuarial present value of benefits based on employee service and compensation as of the measurement date and does not include an assumption about future compensation levels. The following table summarizes the total accumulated benefit obligations, the accumulated benefit obligations and fair value of plan assets for defined benefit pension plans with accumulated benefit obligations in excess of plan assets, and the projected benefit obligation and fair value of plan assets for defined benefit pension plans with projected benefit obligation in excess of plan assets as of December 31, 2025 and December 31, 2024:
| Qualified and Non-qualified Pension Plans | ||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||||
| Accumulated benefit obligation | $ | $ | $ | $ | ||||||||||||||||||||||
| Plans with accumulated benefit obligation in excess of plan assets | ||||||||||||||||||||||||||
| Accumulated benefit obligation | $ | $ | $ | $ | ||||||||||||||||||||||
| Fair value of plan assets | ||||||||||||||||||||||||||
| Plans with projected benefit obligation in excess of plan assets | ||||||||||||||||||||||||||
| Projected benefit obligation | $ | $ | $ | $ | ||||||||||||||||||||||
| Fair value of plan assets | ||||||||||||||||||||||||||
81
Components of Net Periodic Cost and Other Amounts Recognized in Other Comprehensive Income:
The service cost component of defined benefit net periodic benefit cost is recorded in cost of product, cost of software and rentals, selling, general and administrative expenses, and research and development expenses. As discussed in Note 7, the other components of net periodic benefit cost are reflected in other expense (income), net. Components of net periodic benefit cost and other supplemental information for the respective years ended December 31 are as follows:
| Qualified and Non-qualified Pension Benefits | Postretirement Benefits | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | |||||||||||||||||||||||||||||||||||||||||||||||
| Net periodic benefit cost (benefit) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Operating expense | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Service cost | $ | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| Non-operating expense | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interest cost | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expected return on plan assets | ( | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of prior service benefit | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of net actuarial loss | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Settlements, curtailments, and special termination benefits | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total non-operating expense (benefit) | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total net periodic benefit cost (benefit) | $ | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| Other changes in plan assets and benefit obligations recognized in other comprehensive (income) loss | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of transition asset | $ | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of prior service benefit | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net actuarial (gain) loss | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of net actuarial loss | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Settlements, curtailments, and special termination benefits | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total recognized in other comprehensive (income) loss | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total recognized in net periodic benefit cost (benefit) and other comprehensive (income) loss | $ | $ | $ | $ | ( | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||||||||||
Weighted-Average Assumptions Used to Determine Benefit Obligations as of December 31:
| Qualified and Non-qualified Pension Benefits | Postretirement Benefits | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Discount rate | % | % | % | % | % | % | % | % | % | |||||||||||||||||||||||||||||||||||||||||||||||
| Compensation rate increase | % | % | % | % | % | % | N/A | N/A | N/A | |||||||||||||||||||||||||||||||||||||||||||||||
82
Weighted-Average Assumptions Used to Determine Net Cost for Years Ended December 31:
| Qualified and Non-qualified Pension Benefits | Postretirement Benefits | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| United States | International | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Discount rate - service cost | % | % | % | % | % | % | % | % | % | |||||||||||||||||||||||||||||||||||||||||||||||
| Discount rate - interest cost | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expected return on assets | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Compensation rate increase | N/A | N/A | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||
The U.S. postretirement health care benefit plan is a savings account benefit-based plan. The contributions provided by the Company to the health savings accounts increase 3 % per year for employees who retired prior to January 1, 2016 and 1.5 % for employees who retire on or after January 1, 2016. Therefore, the Company does not have material exposure to health care cost inflation related to its retirees.
The Company determines the discount rate to measure plan liabilities as of the December 31 measurement date for the pension and postretirement benefit plans. The discount rate reflects the current rate at which the associated liabilities could be effectively settled. The Company sets its rate to reflect the yield of a portfolio of high quality, fixed-income debt instruments that would produce cash flows sufficient in timing and amount to settle projected future benefits. Actuarial losses in 2025 and gains in 2024 related to projected benefit obligations were primarily the result of changes in discount rates.
The Company measures service cost and interest cost separately using the spot yield curve approach applied to each corresponding obligation. Service costs are determined based on duration-specific spot rates applied to the service cost cash flows. The interest cost calculation is determined by applying duration-specific spot rates to the year-by-year projected benefit payments. The spot yield curve approach does not affect the measurement of the total benefit obligations as the change in service and interest costs offset in the actuarial gains and losses recorded in other comprehensive income.
In 2025, the Company’s assumption for the expected return on plan assets for the primary U.S. qualified pension plan was 7.25 %. The expected return assumption is based on the strategic asset allocation of the plan, long-term capital market return expectations and expected performance from active investment management. As of December 31, 2025, the Company’s 2026 expected long-term rate of return on U.S. plan assets is 7.50 %, which is based on an asset allocation assumption of 10 % global equities, 15 % private equities, 61 % fixed-income securities, and 14 % absolute return investments independent of traditional performance benchmarks, along with positive returns from active investment management. The actual net rate of return on plan assets in 2025 was 8.4 %. Return on assets assumptions for international pension plans are calculated on a plan-by-plan basis using plan asset allocations and expected long-term rate of return assumptions.
In 2026, the Company expects to contribute $22 million to its international pension plans. The Company does not have required minimum cash contributions for its U.S. pension or postretirement plans for 2026. Future contributions will depend on market conditions, interest rates and other factors.
Future Pension and Postretirement Benefit Payments
The following table provides the estimated pension and postretirement benefit payments that are payable from the plans to participants:
| Qualified and Non-qualified Pension Benefits | Postretirement Benefits | |||||||||||||||||||
| (Millions) | United States | International | ||||||||||||||||||
| 2026 Benefit payments | $ | $ | $ | |||||||||||||||||
| 2027 Benefit payments | ||||||||||||||||||||
| 2028 Benefit payments | ||||||||||||||||||||
| 2029 Benefit payments | ||||||||||||||||||||
| 2030 Benefit payments | ||||||||||||||||||||
| Next five years | ||||||||||||||||||||
83
Plan Asset Management
Solventum's investment strategy for its pension and postretirement plans is to manage the funds on a going-concern basis. The primary goal is to meet the obligations as required. The secondary goal is to earn the highest rate of return possible, without jeopardizing its primary goal, and without subjecting the Company to an undue amount of contribution risk. Fund returns are used to help finance present and future obligations to the extent possible within actuarially determined funding limits and tax-determined asset limits, thus reducing the potential need for additional contributions from the Company. The investment strategy has used long duration cash bonds and derivative instruments to offset a significant portion of the interest rate sensitivity of pension liabilities.
Normally, Solventum does not buy or sell any of its own securities as a direct investment for its pension and other postretirement benefit funds. However, due to external investment management of the funds, the plans may indirectly buy, sell or hold Solventum securities. The aggregate amount of Solventum securities are not considered to be material relative to the aggregate fund percentages.
The discussion that follows references the fair value measurements of certain assets in terms of levels 1, 2 and 3. See Note 1 for descriptions of these levels. While the company believes the valuation methods are appropriate and consistent with other market participants, the use of different methodologies or assumptions to determine the fair value of certain financial instruments could result in a different estimate of fair value at the reporting date.
U.S. Pension Plan and Postretirement Benefit Plan Assets
The U.S. pension plan assets and U.S. postretirement plan assets are held in a master trust and are invested with the same investment objectives. In order to achieve the investment objectives, the investment policies include a target strategic asset allocation. The investment policies allow some tolerance around the target in recognition that market fluctuations and illiquidity of some investments may cause the allocation to a specific asset class to vary from the target allocation, potentially for long periods of time. Acceptable ranges have been designed to allow for deviation from strategic targets and to allow for the opportunity for tactical over- and under-weights. The portfolios will normally be rebalanced when the quarter-end asset allocation deviates from acceptable ranges. The allocation is reviewed regularly by the named fiduciary of the plans.
84
The fair values of the assets held by the U.S. pension and postretirement benefit plans by asset class are as follows:
| Fair Value Measurements Using Inputs Considered as | Investments Measured at Net Asset Value* | Fair Value at December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| U.S. Pension Plans | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equities | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed income | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Absolute return | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Private equity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other items to reconcile to fair value of plan assets | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value of plan assets | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Postretirement Benefit Plans | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equities | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed income | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Absolute return | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Private equity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other items to reconcile to fair value of plan assets | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value of plan assets | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
*In accordance with ASC 820-10, certain investments that are measured at fair value using the NAV per share (or its equivalent) as a practical expedient have not been classified in the fair value hierarchy. The NAV is based on the fair value of the underlying assets owned by the fund, minus its liabilities then divided by the number of units outstanding and is determined by the investment manager or custodian of the fund. The fair value amounts presented in this table are intended to permit reconciliation of the fair value hierarchy to the amounts presented in the fair value of plan assets.
Equities consist primarily of mandates in public equity securities managed to various public equity indices. Publicly traded equities are valued at the closing price reported in the active market in which the individual securities are traded. Common collective trusts that invest in equity securities are valued at NAV.
Fixed income investments include domestic government and corporate debt securities, as well as bond futures. The debt securities are valued at the closing price reported if traded on an active market or at yields currently available on comparable securities of issuers with similar credit ratings. Futures are valued at the closing price reported in active market in which the derivative is traded.
Absolute return consists primarily of private partnership interests in hedge funds valued at NAV.
The private equity portfolio consists of partnership interests valued at NAV.
Other items to reconcile to fair value of plan assets as of December 31, 2025 include interest receivables and receivables for investments sold. Other items to reconcile to fair value of plan assets as of December 31, 2024 include interest receivables.
International Pension Plan Assets
Outside the U.S., pension plan assets are typically managed by decentralized fiduciary committees. The disclosure below of asset categories is presented in aggregate for the 13 defined benefit plans in 9 countries, which have plan assets; however, there is significant variation in asset allocation policy from country to country. Local regulations, local funding rules, and local financial and tax considerations are part of the funding and investment allocation process in each country. The Company provides standard funding and investment guidance to all international plans with more focused guidance to the larger plans. Each plan has its own strategic asset allocation. The asset allocations are reviewed periodically and rebalanced when necessary.
85
The fair values of the assets held by the international pension plans by asset class are as follows:
| Fair Value Measurements Using Inputs Considered as | Investments Measured at Net Asset Value* | Fair Value at December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equities | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed income | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Absolute return | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Private equity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other items to reconcile to fair value of plan assets | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value of plan assets | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
*In accordance with ASC 820-10, certain investments that are measured at fair value using the NAV per share (or its equivalent) as a practical expedient have not been classified in the fair value hierarchy. The NAV is based on the fair value of the underlying assets owned by the fund, minus its liabilities, then divided by the number of units outstanding, and is determined by the investment manager or custodian of the fund. The fair value amounts presented in this table are intended to permit reconciliation of the fair value hierarchy to the amounts presented in the fair value of plan assets.
Equities consist primarily of mandates in public equity securities managed to various public equity indices. Publicly traded equities are valued at the closing price reported in the active market in which the individual securities are traded. Commingled funds are valued using the NAV provided by the fund administrator. Assets in the Level 2 category have a quoted market price.
Fixed income investments include domestic and foreign government, and corporate debt securities. The debt securities are valued at the closing price reported if traded on an active market or at yields currently available on comparable securities of issuers with similar credit ratings. Commingled funds are valued using the NAV provided by the fund administrator. Assets in the Level 2 category have a quoted market price.
Absolute return consists of insurance contracts, which are valued using cash surrender values which is the amount the plan would receive if the contract were cashed out at year end, and commingled funds that invest in real estate, which are valued at NAV.
Private equity funds consist of partnership interests in a variety of funds which are valued at NAV as described above.
Other items to reconcile to fair value of plan assets as of December 31, 2025 include payables related to plan assets that are legally due to Buyer as a result of the divestiture of the Purification and Filtration business, which will be transferred to the Buyer in 2026. Other items to reconcile to fair value of plan assets as of December 31, 2024 include a receivable from a 3M-sponsored pension plan that was transferred to the Solventum-sponsored pension plan in 2025.
The balances of and changes in the fair values of the international pension plans’ level 3 assets consist primarily of insurance contracts under the absolute return asset class which were transferred from 3M plans; these investments have few transactions.
Defined Contribution Plans
The Company also sponsors employee savings plans under Section 401(k) of the Internal Revenue Code. These plans are offered to substantially all regular U.S. employees. For eligible employees hired prior to January 1, 2009, employee 401(k) contributions of up to 5 % of eligible compensation are matched in cash at rates of 45 % or 60 %, depending on the plan in which the employee participates. Employees hired on or after January 1, 2009, receive a cash match of 100 % for employee 401(k) contributions of up to 5 % of eligible compensation and receive an employer retirement income account cash contribution of 3 % of the participant’s total eligible compensation. All contributions are invested in a number of investment funds pursuant to employees’ elections. Solventum subsidiaries in various international countries also participate in defined contribution plans. For the year ended December 31, 2025, expense related to the U.S. defined contribution plans was $100 million, and expense related to international defined contribution plans was $31 million. For the year ended December 31, 2024, expense related to the U.S. defined contribution plans was $93 million, and expense related to international defined contribution plans was $24 million. Prior to 2024, employees received employer contributions as part of 3M-sponsored plans.
86
NOTE 11. Derivatives
Cash Flow Hedges - For derivative instruments that are designated and qualify as cash flow hedges, the gain or loss on the derivative is reported as a component of other comprehensive income (loss) and reclassified into earnings in the same period during which the hedged transaction affects earnings. Gains and losses on the derivative representing hedge components excluded from the assessment of effectiveness are recognized in current earnings.
Cash Flow Hedging - Foreign Currency Forward Contracts: The Company enters into foreign exchange forward contracts to hedge against the effect of exchange rate fluctuations on cash flows denominated in foreign currencies. These transactions are designated as cash flow hedges. The settlement or extension of these derivatives will result in reclassifications (from accumulated other comprehensive income) to earnings in the period during which the hedged transactions affect earnings. Solventum may de-designate these cash flow hedge relationships in advance of the occurrence of the forecasted transaction. The portion of gains or losses on the derivative instrument previously included in accumulated other comprehensive income (loss) for de-designated hedges remains in accumulated other comprehensive income (loss) until the forecasted transaction occurs or becomes probable of not occurring. Changes in the value of derivative instruments after de-designation are recorded in earnings. The maximum length of time over which Solventum hedges its exposure to the variability in future cash flows of the forecasted transactions is 24 months.
As of December 31, 2025, the Company had a balance of $8 million associated with the after-tax net unrealized loss associated with cash flow hedging instruments recorded in accumulated other comprehensive income. Of the total after-tax net unrealized balance as of December 31, 2025, Solventum expects to reclassify to earnings approximately $6 million after-tax net unrealized loss over the next 12 months based on exchange rates as of December 31, 2025.
Fair Value Hedges - The Company enters into interest rate swaps to manage its exposure to changes in fair value of the Company's fixed-rate debt. Under these arrangements, the Company agrees to exchange, at specific intervals, the difference between fixed and floating interest amounts calculated by reference to an agreed-upon notional principal amount. Gains and losses associated with interest rate swaps and changes in fair value of the hedged debt are recorded to interest expense in the period they occur.
In December 2025, the Company entered into a $100 million notional fixed-to-floating interest rate swap with a maturity date of March 2033. In January 2026, the Company entered into an additional $100 million notional fixed-to-floating interest rate swap with a maturity date of March 2031. In February 2026, the Company entered into an additional $100 million notional fixed-to-floating interest rate swap with a maturity date of March 2033. These derivatives were designated as fair value hedges of the Company's Senior Notes.
At December 31, 2025, the total notional amount of interest rate swaps designated as fair value hedges was $100 million.
Net Investment Hedges - The Company enters into cross-currency swaps to hedge portions of the Company's investment in foreign operations and manage foreign exchange risk. For instruments that are designated and qualify as hedges of net investments in foreign operations and that meet the effectiveness requirements, the net gains and losses attributable to changes in spot exchange rates are recorded in cumulative translation within other comprehensive income. The remainder of the change in value of such instruments is recorded in earnings. Recognition in earnings of amounts previously recorded in cumulative translation is limited to circumstances such as complete or substantially complete liquidation of the net investment in the foreign operation.
At December 31, 2025, the total notional amount of cross-currency swaps designated as net investment hedges was approximately $1.2 billion.
87
Derivatives Not Designated as Hedging Instruments - Derivatives not designated as hedging instruments include foreign currency contracts to offset, in part, the impacts of changes in value of various non-functional currency denominated items including certain intercompany financing balances.
These derivative instruments are not designated in a hedging relationship; therefore, fair value gains and losses on these contracts are recorded in earnings. The Company does not hold or issue derivative financial instruments for trading purposes.
During the third quarter of 2025, the Company entered into $200 million notional fixed-to-floating forward interest rate swaps and $400 million notional treasury locks in connection with the anticipated closing of the Purification and Filtration sale and planned purchase of a portion of our outstanding registered debt. These instruments were settled during the third quarter of 2025 for a gain of $11 million that was reflected within loss on debt extinguishment, net on the consolidated statements of income.
Statement of Income Location and Impact of Cash Flow Derivative Instruments
The impact to income related to both derivative instruments designated in cash flow hedging relationships and those not designated as hedging instruments for the year ended December 31, 2025 was not material. The impact from derivative instruments designated in cash flow hedging relationships was reflected within cost of sales and the impact from derivatives not designated as hedging instruments was reflected within other expense (income), net on the consolidated statements of income.
Location, Fair Value, and Gross Notional Amounts of Derivative Instruments
The following tables summarize the fair value of Solventum’s derivative instruments and their location in the consolidated balance sheets. Notional amounts below are presented at period end foreign exchange rates.
| Gross Notional Amount | Assets | Liabilities | ||||||||||||||||||||||||||||||||||||||||||||||||
| (Millions) | Location | Fair Value Amount | Location | Fair Value Amount | ||||||||||||||||||||||||||||||||||||||||||||||
| December 31, | December 31, | December 31, | ||||||||||||||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2025 | 2024 | |||||||||||||||||||||||||||||||||||||||||||||
| Derivatives designated as hedging instruments | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency forward contracts | $ | $ | Other current assets | $ | $ | Other current liabilities | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
| Foreign currency forward contracts | Other assets | Other liabilities | ||||||||||||||||||||||||||||||||||||||||||||||||
| Interest rate swaps | Other assets | Other liabilities | ||||||||||||||||||||||||||||||||||||||||||||||||
| Cross-currency swaps | Other assets | Other liabilities | ||||||||||||||||||||||||||||||||||||||||||||||||
| Total derivatives designated as hedging instruments | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Derivatives not designated as hedging instruments | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency forward contracts | $ | $ | Other current assets | $ | $ | Other current liabilities | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
Fair Value Disclosure: The Company’s derivative assets and liabilities within the scope of ASC 815, Derivatives and Hedging, are required to be recorded at fair value. The Company’s derivatives that are recorded at fair value include foreign currency forward contracts and cross-currency swaps. Solventum has determined that these derivatives are considered Level 2 fair value measurements. Solventum determines fair value using observable inputs including foreign currency exchange rates.
Credit Risk and Offsetting of Assets and Liabilities of Derivative Instruments: The Company is exposed to credit loss in the event of nonperformance by counterparties in forward contracts. However, the Company’s risk is limited to the fair value of the instruments. The Company actively monitors its exposure to credit risk through the use of credit approvals and credit limits, and by selecting major international banks and financial institutions as counterparties. Solventum enters into master netting arrangements with counterparties, which may allow each counterparty to net settle amounts owed between a Solventum entity and the counterparty as a result of multiple, separate derivative transactions. The Company does not anticipate nonperformance by any of these counterparties.
Solventum has elected to present the fair value of derivative assets and liabilities within the Company’s consolidated balance sheets on a gross basis even when derivative transactions are subject to master netting arrangements and may otherwise qualify for net presentation. Solventum determined that the impact of the amount of eligible offsetting derivative assets and liabilities was not material if it had elected to offset the asset and liability balances of derivative instruments, netted in accordance with various criteria in the event of default or termination as stipulated by the terms of netting arrangements with each of the counterparties. For each counterparty, if netted, the Company would offset the asset and liability balances of all derivatives at
88
the end of the reporting period based on the Solventum entity that is a party to the transactions. Derivatives not subject to master netting agreements are not eligible for net presentation. For the periods presented, Solventum has not received cash collateral from derivative counterparties.
NOTE 12. Commitments and Contingencies
Legal Proceedings
Solventum is involved in numerous claims and lawsuits, principally in the United States, and regulatory proceedings worldwide. These claims, lawsuits and proceedings relate to matters including, but not limited to, product liability (involving products that the Company now or formerly manufactured and sold, including products made by the Health Care Business Group at 3M), intellectual property, commercial, antitrust, federal healthcare program related laws and regulations, such as the False Claims Act and anti-kickback laws in the United States and other jurisdictions. Unless otherwise stated, Solventum is vigorously defending all such litigation and proceedings. From time to time, Solventum also receives subpoenas, investigative demands or requests for information from various government agencies in the United States and foreign countries. Solventum generally responds in a cooperative, thorough and timely manner. These responses sometimes require time and effort and can result in considerable costs being incurred by the Company. Such requests can also lead to the assertion of claims or the commencement of administrative, civil, or criminal legal proceedings against Solventum and others, as well as to settlements. The outcomes of legal proceedings and regulatory matters are often difficult to predict. Any determination that the Company’s operations or activities are not, or were not, in compliance with applicable laws or regulations could result in the imposition of fines, civil or criminal penalties, and equitable remedies, including disgorgement, suspension or debarment or injunctive relief.
Process for Disclosure and Recording of Liabilities Related to Legal Proceedings
Many lawsuits and claims involve highly complex issues relating to causation, scientific evidence, and alleged actual damages, all of which are otherwise subject to substantial uncertainties. Assessments of lawsuits and claims can involve a series of complex judgments about future events and can rely heavily on estimates and assumptions. The categories of legal proceedings in which the Company is involved may include multiple lawsuits and claims, may be spread across multiple jurisdictions and courts that may handle the lawsuits and claims differently, may involve numerous and different types of plaintiffs, raising claims and legal theories based on specific allegations that may not apply to other matters, and may seek substantial compensatory and, in some cases, punitive, damages. These and other factors contribute to the complexity of these lawsuits and claims and make it difficult for the Company to predict outcomes and make reasonable estimates of any resulting losses. The Company's ability to predict outcomes and make reasonable estimates of potential losses is further influenced by the fact that a resolution of one or more matters within a category of legal proceedings may impact the resolution of other matters in that category in terms of timing, amount of liability, or both.
When making determinations about recording liabilities related to legal proceedings, the Company complies with the requirements of ASC 450, Contingencies, and related guidance, and records liabilities in those instances where it can reasonably estimate the amount of the loss and when the loss is probable. Where the reasonable estimate of the probable loss is a range, the Company records as an accrual in its financial statements the most likely estimate of the loss, or the low end of the range if there is no one best estimate. The Company either discloses the amount of a possible loss or range of loss in excess of established accruals if estimable, or states that such an estimate cannot be made. The Company discloses significant legal proceedings even where liability is not probable or the amount of the liability is not estimable, or both, if the Company believes there is at least a reasonable possibility that a loss may be incurred. Based on experience and developments, the Company reexamines its estimates of probable liabilities and associated expenses and receivables each period, and whether a loss previously determined to not be reasonably estimable and/or not probable is now able to be reasonably estimated or has become probable. Where appropriate, the Company makes additions to or adjustments of its reasonably estimated losses and/or accruals. As a result, the current accruals and/or estimates of loss and the estimates of the potential impact on the Company’s consolidated financial position, results of operations and cash flows for the legal proceedings and claims pending against the Company will likely change over time. During 2025 and 2024, the Company recognized $22 million and $8 million in legal charges, respectively. During 2025 and 2024, the Company made payments of $16 million and $6 million, respectively, related to a legal settlement, which reduced the accrued litigation balance. At December 31, 2025 and December 31, 2024, accrued litigation costs were $31 million and $25 million, respectively.
Because litigation is subject to inherent uncertainties, and unfavorable rulings or developments could occur, the Company may ultimately incur charges substantially in excess of presently recorded liabilities, including with respect to matters for which no accruals are currently recorded because losses are not currently probable and reasonably estimable. Many of the matters described herein are at varying stages, seek an indeterminate amount of damages or seek damages in amounts that the Company believes are not indicative of the ultimate losses that may be incurred. It is not uncommon for claims to be resolved over many years. As a matter progresses, the Company may receive information, through plaintiff demands, through discovery, in the form of reports of purported experts, or in the context of settlement or mediation discussions that purport to quantify an amount of
89
alleged damages, but with which the Company may not agree. Such information may or may not lead the Company to determine that it is able to make a reasonable estimate as to a probable loss or range of loss in connection with a matter. However, even when a loss or range of loss is not probable and reasonably estimable, developments in, or the ultimate resolution of, a matter could be material to the Company and could have a material adverse effect on the Company, its consolidated financial position, results of operations and cash flows. In addition, future adverse rulings or developments, or settlements in, one or more matters could result in future changes to determinations of probable and reasonably estimable losses in other matters.
Process for Disclosure and Recording of Insurance Receivables Related to Legal Proceedings
The Company estimates insurance receivables based on an analysis of the terms of its numerous policies, including their exclusions, pertinent case law interpreting comparable policies, its experience with similar claims, and assessment of the nature of the claim and remaining coverage, and records an amount it has concluded is recognizable and expects to receive in light of the loss recovery and/or gain contingency models under ASC 450, ASC 610-30, and related guidance. For those insured legal proceedings for which the Company has recorded an accrued liability in its financial statements, the Company also records receivables for the amount of insurance that it concludes as recognizable from the Company’s insurance program. For those insured matters for which the Company has not recorded an accrued liability because the liability is not probable or the amount of the liability is not estimable, or both, but for which the Company has incurred an expense in defending itself, the Company records receivables for the amount of insurance that it concludes as recognizable for the expense incurred.
Product Liability Litigation
The following sections first describe the significant legal proceedings in which the Company is involved, and then describe the liabilities, if any, the Company has accrued relating to its significant legal proceedings.
3M is a named defendant in over 8,400 lawsuits in the United States and one Canadian putative class action with a single named plaintiff, alleging that they underwent various joint arthroplasty, cardiovascular, and other surgeries and later developed surgical site infections due to the use of the Bair Hugger patient warming system. Under the terms of the Separation and Distribution Agreement by and between Solventum and 3M (the "Separation and Distribution Agreement"), Solventum has agreed to indemnify 3M for uninsured liabilities related to the Bair Hugger patient warming system, to manage the litigation, and pay for legal expenses.
The U.S. Judicial Panel on Multidistrict Litigation ("JPML") has consolidated all cases pending in federal courts to the U.S. District Court for the District of Minnesota to be managed in a multi-district litigation ("MDL") proceeding. In July 2019, the court excluded several of the plaintiffs’ causation experts, and granted summary judgment for 3M in all cases pending at that time in the MDL; however, those decisions were subsequently reversed by the U.S. Court of Appeals for the Eighth Circuit. The parties are actively litigating several MDL bellwether and state court cases, with trials anticipated in 2026.
In addition to the federal MDL cases, there are eight state court personal injury cases relating to the Bair Hugger patient warming systems, including a multi-plaintiff case of amputees in Ramsey County, Minnesota. Additionally, a putative class action has been filed in Ramsey County, Minnesota, seeking economic damages for the use of the Bair Hugger system in knee and hip replacement surgeries involving medically obese people in Minnesota from May 2017 to the present.
In June 2016, 3M was served with a putative class action filed in the Ontario Superior Court of Justice for all Canadian residents who underwent various joint arthroplasty, cardiovascular, and other surgeries and later developed surgical site infections that the representative plaintiff claims were due to the use of the Bair Hugger patient warming system. The representative plaintiff seeks relief (including punitive damages) under Canadian law based on theories similar to those asserted in the MDL.
For product liability litigation matters described in this section for which a liability has been recorded, the amount recorded is included in the disclosed amounts in the preceding "Process for Disclosure and Recording of Liabilities Related to Legal Proceedings" section and is not material to the Company’s results of operations or financial condition. In addition, the Company is not able to estimate a possible loss or range of possible loss in excess of the recorded liability at this time.
Federal False Claims Act/Qui Tam Litigation
In October 2019, 3M acquired Acelity, Inc. and its KCI subsidiaries, including Kinetic Concepts, Inc. and KCI USA, Inc.
In 2008, two former employees filed qui tam actions against Kinetic Concepts, Inc. and KCI USA, Inc. (collectively, the “KCI Defendants”) alleging that the KCI Defendants violated the federal False Claims Act by submitting false or fraudulent claims to federal healthcare programs related to billing for 3M V.A.C. Therapy. One qui tam action (the Godecke case) was dismissed in January 2022.
90
In the remaining action (the Hartpence case), the litigation remained in a pre-trial stage through July 2023 when the parties agreed to mediate the matter. The parties updated the court periodically during their mediation and subsequent negotiation of an agreement to settle the matter. The parties entered into an agreement resolving the matter on May 15, 2025. On July 2, 2025, pursuant to that agreement, the relator-plaintiff filed a joint stipulation of voluntary dismissal, dismissing the matter with prejudice as to the relator-plaintiff and without prejudice as to the United States. On July 3, 2025, the court entered an order dismissing the matter consistent with the parties’ stipulation.
Warranties/Guarantees
The Company had approximately $82 million and $40 million in bank guarantees, surety bonds, and other similar instruments issued and outstanding at December 31, 2025 and 2024, respectively. These instruments are utilized in connection with normal business activities. Furthermore, the Company does not disclose information on its product warranties, as management considers the balance immaterial to its consolidated results of operations and financial condition.
NOTE 13. Leases
Solventum's lease arrangements include both operating and finance leases. Amounts associated with finance leases were not material to the consolidated financial statements for all periods presented.
During 2024, Solventum entered into a finance lease arrangement through April 2046 for the future location of the Company's principal office in Eagan, Minnesota. The new location will include office, R&D and manufacturing space. The lease will commence on the date that construction is complete and control of the location is transferred to the Company, which is anticipated to be the first quarter of 2026. The Company will record approximately $200 million of finance lease right-of-use assets with corresponding finance lease liabilities upon commencement.
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Operating lease cost | $ | $ | $ | |||||||||||||||||
Variable lease costs, short-term lease cost and income related to sub-lease activity is immaterial for the Company.
Supplemental balance sheet, lease term and discount rate information related to operating leases is as follows:
| December 31, | ||||||||||||||||||||
| (Millions, unless noted) | Location on face of Balance Sheet | 2025 | 2024 | |||||||||||||||||
| Operating leases: | ||||||||||||||||||||
| Operating lease right of use assets | $ | $ | ||||||||||||||||||
| Current operating lease liabilities | ||||||||||||||||||||
| Noncurrent operating lease liabilities | ||||||||||||||||||||
| $ | $ | |||||||||||||||||||
| Weighted average remaining lease term (in years): | ||||||||||||||||||||
| Weighted average discount rate: | % | % | ||||||||||||||||||
Supplemental cash flow information related to operating lease is as follows:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Cash paid for amounts included in the measurement of lease liabilities: | $ | $ | $ | |||||||||||||||||
Right of use assets obtained in exchange for operating lease liabilities: | ||||||||||||||||||||
There was no sale-leaseback activity for the periods presented.
91
Maturities of operating leases as of December 31, 2025 are as follows:
| (Millions) | ||||||||
| 2026 | $ | |||||||
| 2027 | ||||||||
| 2028 | ||||||||
| 2029 | ||||||||
| 2030 | ||||||||
| After 2030 | ||||||||
Total expected lease payments | ||||||||
| Less: Amounts representing interest | ( | |||||||
| Present value of future minimum lease payments | $ | |||||||
NOTE 14. Restructuring
Transform for the Future
In November 2025, the Company approved its new multiyear 'Transform for the Future' global initiative (the "Program") to further enable its long-term growth strategy and ensure it's best positioned to compete-to-win in a rapidly changing healthcare environment. Designed to transform the cost structure, enhance operational efficiency, and reposition for profitable growth, the primary activities of the Program include operating structure optimization and workforce reorganization, procurement and cost management, supply chain, manufacturing and global footprint optimization, and streamlining systems and increased automation to improve operational efficiency. Once fully implemented, the four year Program is expected to generate approximately $500 million in annual cost savings, a portion of which will be reinvested in strategic growth initiatives. The Company anticipates cumulative pretax costs related to the Program will be approximately $500 million.
The related restructuring charges for periods presented were recorded in the consolidated statements of income as follows:
| Year ended December 31, | ||||||||
| (Millions) | 2025 | |||||||
| $ | ||||||||
| Total operating income impact | $ | |||||||
Restructuring actions, including cash and non-cash impacts, are as follows:
| (Millions) | Employee Termination Benefits | Other Restructuring-related | Total | |||||||||||||||||
| Expense incurred in 2025 | $ | $ | $ | |||||||||||||||||
| Non-cash changes | ( | ( | ||||||||||||||||||
| Cash payments | ( | ( | ( | |||||||||||||||||
| Accrued liabilities as of December 31, 2025 | $ | $ | $ | |||||||||||||||||
Other includes charges associated with salaries and wages of employees fully dedicated to the Program and consulting service fees. All Program charges were recognized within Corporate and are not included within business segment results.
92
Solventum Way
In the fourth quarter of 2024, the Company announced its Solventum Way restructuring program, which is a reorganization designed to establish a more flexible and decentralized structure, create headroom to invest for growth and an operating model that enhances margins over time. The actions under the Solventum Way restructuring program were substantially complete as of December 31, 2025.
The related restructuring charges for periods presented were recorded in the consolidated statements of income as follows:
| Year ended December 31 | ||||||||||||||
| (Millions) | 2025 | 2024 | ||||||||||||
| $ | $ | |||||||||||||
| Total operating income impact | $ | $ | ||||||||||||
Restructuring actions, including cash and non-cash impacts, are as follows:
| (Millions) | Employee Termination Benefits | Other Restructuring-related | Total | |||||||||||||||||
| Expense incurred in 2024 | $ | $ | $ | |||||||||||||||||
| Non-cash changes | ( | ( | ||||||||||||||||||
| Cash payments | ( | ( | ||||||||||||||||||
| Accrued liabilities as of December 31, 2024 | $ | $ | $ | |||||||||||||||||
| Expense incurred in 2025 | $ | $ | ( | $ | ||||||||||||||||
| Non-cash changes | ( | ( | ||||||||||||||||||
| Cash payments | ( | ( | ( | |||||||||||||||||
| Accrued liabilities as of December 31, 2025 | $ | $ | $ | |||||||||||||||||
Other primarily includes charges associated with asset write-offs and other contractual third party termination costs. All program charges were recognized within Corporate and are not included within business segment results.
NOTE 15. Earnings Per Share
Prior to the completion of the Spin-Off, the Company had no 172,709,505
The dilutive effect of outstanding stock options, restricted stock units ("RSUs") and performance share units ("PSUs") is reflected in the calculation of earnings per share using the treasury stock method. Diluted earnings per share excludes certain shares issuable under stock-based compensation plans because the effect would have been antidilutive.
The computations for basic and diluted earnings per share are as follows:
93
| Year ended December 31, | ||||||||||||||||||||
| (Millions, except per share amounts) | 2025 | 2024 | 2023 | |||||||||||||||||
| Numerator: | ||||||||||||||||||||
| Net income | $ | $ | $ | |||||||||||||||||
| Denominator: | ||||||||||||||||||||
Weighted average common shares outstanding – basic | ||||||||||||||||||||
| Dilution associated with stock-based compensation plans | ||||||||||||||||||||
| Weighted average common shares outstanding – diluted | ||||||||||||||||||||
| Basic earnings per share | $ | $ | $ | |||||||||||||||||
| Diluted earnings per share | $ | $ | $ | |||||||||||||||||
| Antidilutive shares | ||||||||||||||||||||
NOTE 16. Stock-Based Compensation
Prior to the Spin-Off, certain eligible employees were awarded incentive stock options, non-qualified stock options, stock appreciation rights, RSUs, and PSUs under 3M's Amended and Restated 2016 Long-Term Incentive Plan ("3M Company Plan"). Stock-based compensation granted pursuant to the 3M Company Plan was based on 3M’s common stock.
In March 2024, 3M's Board of Directors approved the 2024 Long-Term Incentive Plan ("Solventum 2024 Plan") providing for the grant of incentive stock options, non-qualified stock options, stock appreciation rights, RSUs, PSUs, and other stock or cash-based awards to eligible employees and non-employee directors. Stock-based compensation granted pursuant to the Solventum 2024 Plan is based on Solventum's common stock. The maximum shares that can be issued under the Solventum 2024 Plan is 18.4 million shares, which represents the sum of 13.0 million shares plus 5.4 million shares related to awards granted under the 3M Company Plan for Solventum employees as described below. As of December 31, 2025, the remaining shares available for future grants under the Solventum 2024 Plan was approximately 8.0 million shares.
In connection with the Spin-Off, all awards granted under the 3M Company Plan for Solventum employees were converted into equivalent awards under the Solventum 2024 Plan. The stock award modification at Spin-Off resulted in incremental compensation cost of $22 million, of which $20 million was recognized during the year ended December 31, 2024, and $2 million was recognized during the year ended December 31, 2025.
Solventum grants annual stock-based compensation awards to certain employees and non-employee directors. In 2025, the annual grant occurred in March and included grants of RSUs and PSUs. The Company makes other minor grants of RSUs during the year. In 2024, the annual grant occurred in May after the Spin-Off and included grants of RSUs and PSUs. Additionally, in 2024, the Company awarded one-time founders' RSUs and inducement PSUs to certain employees and non-employee directors.
94
Stock-Based Compensation Expense
Amounts recognized in the consolidated financial statements related to stock-based compensation awards, including stock options, RSUs, and PSUs, are provided in the following table. Total stock-based compensation expense recognized in cost of product and cost of software and rentals has been combined in the table below within Cost of sales. Capitalized stock-based compensation amounts were not material.
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Cost of sales | $ | $ | $ | |||||||||||||||||
| Selling, general and administrative expenses | ||||||||||||||||||||
| Research and development expenses | ||||||||||||||||||||
| Stock-based compensation expenses | ||||||||||||||||||||
| Income tax benefits (expense) | ( | ( | ( | |||||||||||||||||
Stock-based compensation expenses, net of tax | $ | $ | $ | |||||||||||||||||
Restricted Stock Units
RSUs contain service-only vesting conditions. The fair value of RSUs is based upon Solventum's closing stock price on the grant date, and the awards generally vest over periods ranging from to three years from the grant date assuming continued employment. The following table summarizes the RSU activity:
| (Units in thousands) | Number of Shares | Weighted Average Grant Date Fair Value | ||||||||||||
Non-vested as of December 31, 2024 | $ | |||||||||||||
| Granted | ||||||||||||||
| Vested | ( | |||||||||||||
| Forfeited | ( | |||||||||||||
Non-vested as of December 31, 2025 | $ | |||||||||||||
As of December 31, 2025, there was $92 million of compensation expense that has yet to be recognized related to non-vested RSUs. This expense is expected to be recognized over the remaining weighted-average service period of 22 months.
The following table summarizes additional information relative to RSUs for the respective years. There were no Solventum equity-based awards prior to the Spin-Off:
| (Millions, except per-share amounts) | 2025 | 2024 | 2023 | |||||||||||||||||
| Weighted average grant date fair value of RSUs granted (per unit) | $ | $ | $ | |||||||||||||||||
| Total fair value of RSUs vested | ||||||||||||||||||||
| Tax benefit realized related to RSUs vested | $ | $ | $ | |||||||||||||||||
Performance Share Units
The Company grants PSUs to members of its executive management team. PSUs vest upon completion of the requisite service period of three years , or one year for employees who meet the retirement criteria, and upon the achievement of certain performance metrics or market conditions. Performance metrics relate to Company performance against financial targets related to constant currency revenue and adjusted earnings per share. The market condition relates to Solventum's total shareholder return relative to a selected industry peer group. Performance is measured over three years , and the number of shares of common stock that will vest at the end of the performance period is between 0 % to 200 % of the target number of PSUs granted based upon actual performance. The fair value of the performance vesting conditions is based upon Solventum's closing stock price on the grant date. The fair value of the market vesting condition is estimated using a Monte Carlo simulation.
In 2024, the Talent Committee of the Board of Directors approved PSU awards for members of the executive management team; however, the cumulative three-year performance metrics for these awards were not established. The Company began accruing the compensation cost related to these awards on the service inception date. In 2025, the grant-date fair value of these awards was determined upon the approval of the cumulative three-year performance metrics. The 2025 annual PSUs were also granted in 2025. The following table summarizes the PSU activity; the granted number of shares represents the shares that will vest based upon attainment of the performance and market conditions at target (i.e. 100%):
95
| (Units in thousands) | Number of Shares | Weighted Average Grant Date Fair Value | ||||||||||||
Non-vested as of December 31, 2024 | $ | |||||||||||||
| Granted | ||||||||||||||
| Vested | ||||||||||||||
| Forfeited | ( | |||||||||||||
Non-vested as of December 31, 2025 | $ | |||||||||||||
As of December 31, 2025, there was $53 million of compensation expense that has yet to be recognized related to PSUs. This expense is expected to be recognized over the remaining weighted-average service period of 19 months.
Stock Options
The following table summarizes stock option activity:
| (Options in thousands) | Number of Options | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term (months) | Aggregate Intrinsic Value (millions) | ||||||||||||||||||||||
Outstanding as of December 31, 2024 | $ | |||||||||||||||||||||||||
| Granted | ||||||||||||||||||||||||||
| Exercised | ( | |||||||||||||||||||||||||
| Forfeited | ( | |||||||||||||||||||||||||
| Expired | ( | |||||||||||||||||||||||||
Outstanding as of December 31, 2025 | $ | |||||||||||||||||||||||||
Exercisable as of December 31, 2025 | ||||||||||||||||||||||||||
| Expected to vest | $ | $ | ||||||||||||||||||||||||
Stock options outstanding and exercisable in the table above relate to the awards previously granted under 3M Company. The outstanding stock options vest over a period of three years from the grant date and expire ten years from the grant date. As of December 31, 2025, there was $0.1 million of compensation expense that has yet to be recognized related to non-vested stock options. This expense is expected to be recognized over the remaining weighted-average service period of 1 month.
NOTE 17. Related Parties
Related Party Transactions Prior to Spin-Off
Prior to the Spin-Off, the Company participated in centralized 3M treasury programs. This arrangement was not reflective of the manner in which the Company would have financed its operations had it been a standalone business separate from 3M during the periods presented prior to April 1, 2024. All adjustments relating to certain transactions among the Company and 3M, which include the transfer of the balance of cash to and from 3M, transfer of the balance of cash held in centralized cash management arrangements to and from 3M, and pushdown of all costs of doing business that were paid on behalf of the Company by 3M, are excluded from the asset and liability balances in the consolidated balance sheets and have instead been reported within Net parent investment as a component of equity.
Corporate Allocations
The consolidated statements of income for periods prior to April 1, 2024 include general corporate expenses of 3M for services provided by 3M for certain corporate and shared service functions that were provided on a centralized basis, including the use of shared assets. Expenses had been included on a direct usage basis where costs were specifically identifiable to Solventum or allocated based on the Company’s pro rata proportion of 3M's revenue.
Management believes that the expense allocations were determined on a basis that was a reasonable reflection of the utilization of services provided for or the benefit received by the Company during each of the periods presented prior to April 1, 2024. The amounts that would have been incurred on a standalone basis could materially differ from the amounts allocated. Management does not believe, however, that it is practicable to estimate what these expenses would have been had the Company operated as an independent entity, including any expenses associated with obtaining any of these services from unaffiliated entities. There was no expense allocation activity from 3M after April 1, 2024.
96
3M expense allocations were recorded in the consolidated statements of income within the following captions:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Costs of product | $ | $ | $ | |||||||||||||||||
| Costs of software and rentals | ||||||||||||||||||||
| Selling, general and administrative expenses | ||||||||||||||||||||
| Research and development expenses | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
Related Party Transactions After Spin-Off
Separation and Distribution Agreement and Other Related Party Transactions with 3M
In connection with the Spin-Off on April 1, 2024, the Company entered into or adopted several agreements that provide a framework for Solventum's relationship with 3M after the separation and distribution. Below is a summary of activity between Solventum and 3M for the periods presented:
•Separation related adjustments, which are the net impact of certain assets and liabilities that were retained by 3M and those that were transferred to Solventum as of March 31, 2024 , resulted in a decrease to net assets and total equity of $1.1 billion in the second quarter of 2024. This activity was reflected in the "Net transfers to 3M" line item of the consolidated statements of changes in equity. The impact on net assets primarily represents liabilities payable to 3M for services received prior to Spin-Off as well as cash and accounts receivable retained by 3M.
•Transition agreement expenses for the years ended December 31, 2025 and 2024 were $534 million and $369 million, respectively, and are related to services received under transition agreements between the Company and 3M and its affiliates. These expenses are reflected in cost of product and operating expense (which is comprised of selling, general and administrative and research and development expenses) on the Company's consolidated statements of income.
•Master Supply Agreements - The Company recognized revenue and cost of sales associated with products sold to 3M of $78 million and $58 million, respectively, for the year ended December 31, 2025 and $50 million and $37 million, respectively, for the year ended December 31, 2024. Cost of product related to purchases from 3M under the master supply agreements was $241 million and $128 million for the years ended December 31, 2025 and 2024, respectively.
Related Party Transactions
The Company had the following transactions with 3M and its affiliates, primarily in connection with the transition and master supply agreements, reported in the Company’s consolidated financial statements:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net sales of product | $ | $ | $ | |||||||||||||||||
| Cost of product | ||||||||||||||||||||
| Selling, general and administrative expenses | ||||||||||||||||||||
Research and development expenses | ||||||||||||||||||||
Current amounts due from and due to 3M under various agreements described above are recognized within the due from related parties and due to related parties, as applicable, in the consolidated financial statements. Non-current amounts due to 3M in connection with the transition distribution services agreement were approximately $0 and $170 million at December 31, 2025 and 2024 and were recognized in other liabilities in the consolidated balance sheets.
Net Parent Investment
Net transfers to 3M are included within Net parent investment in the consolidated statements of changes in equity and within financing activities in the consolidated statements of cash flows and represent the net effect of transactions between the Company and 3M.
97
The reconciliation of net transfers to 3M between the consolidated changes in equity and the consolidated statements of cash flows are as follows:
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Net transfers from (to) 3M on the consolidated changes in equity | $ | ( | $ | ( | $ | ( | ||||||||||||||
| Stock compensation expense | ( | ( | ||||||||||||||||||
| Multiemployer pension expense | ( | ( | ||||||||||||||||||
| Net balances transferred from 3M | ||||||||||||||||||||
| Net transfers from (to) 3M on the consolidated statements of cash flows | $ | ( | $ | ( | $ | ( | ||||||||||||||
NOTE 18. Business Segments
Operating segments include components of an enterprise where separate financial information is available that is evaluated regularly by the Company’s Chief Operating Decision Maker ("CODM") for the purpose of assessing performance and allocating resources. The Company’s CODM is its Chief Executive Officer. The primary profitability measurement used by the CODM to review segment operating results is segment operating income. The CODM uses segment operating income to allocate resources during the strategic planning process and then holds the segments accountable to the resourcing decisions during the annual budgeting process. The CODM does not use asset information by segment to evaluate reportable segments as the CODM does not receive discrete asset information by segment. Beginning in third quarter 2025, as a result of the sale of the Purification and Filtration business, the Company’s operating activities are managed primarily through the following reportable segments: MedSurg, Dental Solutions, and Health Information Systems. There have been no changes to the composition of or to financial information reported within each of these reportable segments. These segments have been identified based on the nature of the products sold and how the Company manages its operations. Transactions among reportable segments are recorded at cost. No operating segments have been aggregated to form reportable segments.
All Other includes the Water Business, which was previously reported within the Purification and Filtration business segment. The Water Business results have been reclassified for comparability within All Other for all historical periods. All Other also includes sales and cost of sales related to our supply agreements with 3M and other supply agreements assumed by the Company at Spin-Off related to legacy 3M businesses, which were historically included in Corporate and Unallocated.
Certain items are maintained at the corporate level and not allocated to the segments ("Corporate and Unallocated"). Corporate and Unallocated primarily includes amortization of acquired intangible assets, restructuring and related charges, timing related benefits or costs associated with capitalized manufacturing variances, charges and recoveries related to certain litigation, transaction and employee retention costs related to the acquisition of Acera, and gains on sale of businesses. In addition, Corporate and Unallocated includes Spin-Off and separation related costs. Spin-Off and separation related costs include any costs incurred as part of our separation from 3M and costs to setup operations as a standalone company, including system implementations, manufacturing relocations, legal entity separations, certain equity awards granted as part of the Spin-Off, profit mark-ups on transition service arrangements with 3M and other one-time costs. Corporate and Unallocated also includes income and costs related to transition service agreements entered into in connection with the sale of the Purification and Filtration business.
Because Corporate and Unallocated includes a variety of miscellaneous items, it is subject to fluctuation on a quarterly and annual basis. Business segment operating income is reconciled to total operating income and pre-tax income below.
Consistent accounting policies have been applied on a consolidated basis as well as by all segments for all reporting periods.
Business Segments
We are organized into three reportable business segments that are aligned with the markets we serve.
MedSurg Provider of a broad range of innovative, advanced wound care and surgical solutions that are intended to accelerate healing, prevent complications and lower the total cost of care globally.
Dental Solutions Provider of a comprehensive suite of dental and orthodontic products that span the life of the tooth, which are intended to address clinical needs in prevention, restoration, replacement, and malocclusion correction.
Health Information Systems Provider of software solutions and services that are designed to create more time for clinicians to care for patients, improve accuracy in healthcare reimbursement, and support the shift to value-based care.
98
Business Segment Information and Disaggregated Net Sales
| Year ended December 31, | ||||||||||||||||||||
Net Sales (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| Advanced Wound Care | $ | $ | $ | |||||||||||||||||
| Infection Prevention and Surgical Solutions | ||||||||||||||||||||
| MedSurg | ||||||||||||||||||||
| Dental Solutions | ||||||||||||||||||||
| Health Information Systems | ||||||||||||||||||||
| Total reportable segment net sales | ||||||||||||||||||||
| Purification and Filtration | ||||||||||||||||||||
| All Other | ||||||||||||||||||||
| Total net sales | $ | $ | $ | |||||||||||||||||
| Year ended December 31, | ||||||||||||||||||||
| Cost of Sales (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| MedSurg | $ | $ | $ | |||||||||||||||||
| Dental Solutions | ||||||||||||||||||||
| Health Information Systems | ||||||||||||||||||||
| Year ended December 31, | ||||||||||||||||||||
| Operating Expenses (Millions)* | 2025 | 2024 | 2023 | |||||||||||||||||
| MedSurg | $ | $ | $ | |||||||||||||||||
| Dental Solutions | ||||||||||||||||||||
| Health Information Systems | ||||||||||||||||||||
| * Operating expenses are comprised of selling, general and administrative expenses and research and development expenses as shown on the consolidated statements of income. | ||||||||||||||||||||
| Year ended December 31, | ||||||||||||||||||||
Operating Performance (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| MedSurg | $ | $ | $ | |||||||||||||||||
| Dental Solutions | ||||||||||||||||||||
| Health Information Systems | ||||||||||||||||||||
| Total reportable segment operating income | ||||||||||||||||||||
| Purification and Filtration | ||||||||||||||||||||
| All Other | ||||||||||||||||||||
| Amortization expense | ( | ( | ( | |||||||||||||||||
| Corporate and Unallocated | ( | ( | ||||||||||||||||||
| Total operating income | ||||||||||||||||||||
| Interest expense, net | ||||||||||||||||||||
| Loss on extinguishment of debt, net | ||||||||||||||||||||
| Other expense/(income), net | ||||||||||||||||||||
| Income before income taxes | $ | $ | $ | |||||||||||||||||
The following table represents the depreciation amounts reported within the business segment operating income for our reportable segments. The amounts reflected below include both depreciation on property, plant and equipment and equipment held for use. These amounts are included within Cost of Sales and Operating Expenses disclosed in the segment table above.
| Year ended December 31, | ||||||||||||||||||||
| (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| MedSurg | $ | $ | $ | |||||||||||||||||
| Dental Solutions | ||||||||||||||||||||
| Health Information Systems | ||||||||||||||||||||
99
Geographic Information:
Sales are generally reported within the geographic area that originated the invoice to the Company's customer.
| Year ended December 31, | ||||||||||||||||||||
| Net Sales (Millions) | 2025 | 2024 | 2023 | |||||||||||||||||
| United States | $ | $ | $ | |||||||||||||||||
| International | ||||||||||||||||||||
| Worldwide | $ | $ | $ | |||||||||||||||||
Long-lived assets include property, plant, and equipment, equipment rented to customers, as well as operating lease right-of-use assets. The following table presents long-lived assets based on the physical location of those assets.
| December 31, | ||||||||||||||
| Long Lived Assets (Millions) | 2025 | 2024 | ||||||||||||
| United States | $ | $ | ||||||||||||
| Germany | ||||||||||||||
| Other Countries | ||||||||||||||
| Worldwide | $ | $ | ||||||||||||
100
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Processes
The Company carried out an evaluation, under the supervision and with the participation of its management, including the Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of the Company’s "disclosure controls and procedures" (as defined in the Exchange Act Rule 13a-15(e)) as of the end of the period covered by this report. Based upon that evaluation, the Chief Executive Officer and Chief Financial Officer concluded that the Company’s disclosure controls and procedures are effective as of December 31, 2025.
Management's Responsibility for Financial Reporting
Management is responsible for the integrity and objectivity of the financial information included in this report. The financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America. Where necessary, the financial statements reflect estimates based on management’s judgment.
Management has established and maintains a system of internal control over financial reporting for the Company and its subsidiaries. This system and its established accounting procedures and related controls are designed to provide reasonable assurance that assets are safeguarded, that the books and records properly reflect all transactions, that policies and procedures are implemented by qualified personnel, and that published financial statements are properly prepared and fairly presented. The Company’s system of internal control over financial reporting is supported by widely communicated written policies, including business conduct policies, which are designed to require all employees to maintain high ethical standards in the conduct of Company affairs. Internal auditors continually review the accounting and control system.
Management's Report on Internal Control Over Financial Reporting
The Company’s management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rules 13a-15(f) and 15-d-15(f) under the Exchange Act. Management conducted an assessment of the Company’s internal control over financial reporting based on the framework established by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control — Integrated Framework (2013). Management’s assessment of the effectiveness of the Company’s internal control over financial reporting as of December 31, 2025 excluded Acera, a wholly-owned subsidiary, which was acquired by the Company in a purchase business combination in December 2025. Total assets and total net sales of Acera represent less than 1 percent of the related consolidated financial statement amounts as of December 31, 2025. Companies are allowed to exclude acquisitions from their assessment of internal control over financial reporting during the year of acquisition while integrating the acquired company under guidance established by the Securities and Exchange Commission Staff. Based on the assessment, management concluded that, as of December 31, 2025, the Company’s internal control over financial reporting is effective.
The effectiveness of the Company’s internal control over financial reporting as of December 31, 2025 has been audited by PricewaterhouseCoopers LLP, an independent registered public accounting firm, as stated in their report which is included herein.
Changes in Internal Control Over Financial Reporting
There was no change in the Company’s internal control over financial reporting that occurred during the Company’s most recently completed fiscal quarter that has materially affected, or is reasonably likely to materially affect, the Company’s internal control over financial reporting.
101
Item 9B. Other Information
Insider Trading Arrangements and Policies
During the quarter ended December 31, 2025, no director or officer of the Company adopted terminated
Time Sharing Agreement between Solventum Corporation and Wayde McMillan
On February 24, 2026, the Company entered into an aircraft time sharing agreement with Wayde McMillan, the Company’s Chief Financial Officer, allowing Mr. McMillan to reimburse the Company for costs associated with his non-business use of company aircraft. The Talent Committee approved Mr. McMillan’s non-business use of company aircraft for up to 30 hours per year, subject to reimbursement of the aggregate incremental costs of such use, to provide time efficiencies, a confidential work environment and enhanced security for the CFO, which use is subject to the Company’s aircraft use policies. A copy of the time sharing agreement is filed as Exhibit 10.28 herewith.
Availability of Information
Solventum's website address is www.solventum.com. Investors and others should note that the Company announces material information to its investors using SEC filings, press releases, its investor relations website, public conference calls and webcasts. The Company uses these channels to communicate with investors, customers and the public about the Company, its products and other issues. The information on, or that may be accessed through, Solventum's website is not incorporated by reference into this Annual Report on Form 10-K and should not be considered a part of this Annual Report on Form 10-K.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable
102
PART III
Documents Incorporated by Reference
In response to Part III, Items 10, 11, 12, 13 and 14, parts of the Company’s definitive proxy statement (to be filed pursuant to Schedule 14A within 120 days after Solventum’s fiscal year-end of December 31, 2025) for its annual meeting to be held on May 15, 2026, are incorporated by reference in this Form 10-K.
Item 10. Directors, Executive Officers and Corporate Governance
Directors
The information relating to Solventum’s Board of Directors and nominees is set forth under the caption "Proposal 1: Election of Class II Directors" in Solventum’s proxy statement for its annual meeting of stockholders to be held on May 15, 2026 (the "Solventum Proxy Statement") and is incorporated by reference herein.
Executive Officers
Information about Solventum’s executive officers is included in Part I, Item 1 of this Annual Report on Form 10-K and incorporated by reference herein.
Corporate Governance
The information required by Items 407(c)(3), (d)(4) and (d)(5) of Regulation S-K is contained under the captions "Corporate governance — Director independence," "Board composition and leadership structure — Board committees," and "Proposal 1: Election of Class II Directors — Directors’ skills, backgrounds and expertise," of the Solventum Proxy Statement and such information is incorporated by reference herein.
Code of Ethics and Insider Trading Policy
All of our employees, including our Chief Executive Officer, Chief Financial Officer and Controller and Chief Accounting Officer, are required to abide by Solventum’s Code of Conduct to ensure that our business is conducted in a consistently legal and ethical manner. Solventum has posted the text of the Code of Conduct on its website (https://www.solventum.com/en-us/home/our-company/ethics-compliance/#code-conduct). At the same website, any future amendments to the code of ethics will also be posted. Any person may request a copy of the code of ethics, at no cost, by writing to us at the following address:
| Solventum Corporation | ||||||||
| 2510 Conway Ave | ||||||||
| St. Paul, MN 55144 | ||||||||
| Attention: Chief Ethics & Compliance Officer | ||||||||
The information required by Item 408(b) of Regulation S-K is contained under the caption "Compensation discussion and analysis — Trading policy and prohibition on pledging and hedging" of the Solventum Proxy Statement and such information is incorporated by reference herein.
Item 11. Executive Compensation
The information required by Item 402 of Regulation S-K is contained under the captions "Compensation discussion and analysis," "Compensation tables," and "Proposal 1: Election of Class II Directors" of the Solventum Proxy Statement. Such information is incorporated by reference.
The information required by Items 407(e)(4) and (e)(5) of Regulation S-K is contained under the captions "Compensation discussion and analysis — Talent committee report" and "Corporate governance — Compensation committee interlocks and insider participation" of the Solventum Proxy Statement. Such information (other than the Talent Committee Report, which shall not be deemed to be "filed") is incorporated by reference.
103
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required under Item 403 of Regulation S-K is incorporated by reference to the Solventum Proxy Statement under the caption "Security ownership of certain beneficial owners" and the information required under Item 201(d) of Regulation S-K is incorporated by reference to the Solventum Proxy Statement under the caption "Equity compensation plan information".
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by Item 404(b) of Regulation S-K is contained under the captions "Corporate governance — Certain relationships and related person transactions," and the information required by Item 407(a) of Regulation S-K is contained under the caption "Corporate governance — Director independence" in the Solventum Proxy Statement. Such information is incorporated by reference.
Item 14. Principal Accounting Fees and Services
The information relating to principal accounting fees and services is set forth in the section entitled "Proposal 3: Ratification of PwC as Auditor" in the Solventum Proxy Statement and such information is incorporated by reference herein.
104
PART IV
Item 15. Exhibits, Financial Statement Schedules
(a) (1) Financial Statements. The consolidated financial statements filed as part of this report are listed in the index to financial statements at the beginning of this document.
(a) (2) Financial Statement Schedules. Financial statement schedules are omitted because of the absence of the conditions under which they are required or because the required information is included in the Consolidated Financial Statements or the notes thereto. The financial statements of unconsolidated subsidiaries are omitted because, considered in the aggregate, they would not constitute a significant subsidiary.
(a) (3) Exhibits. The exhibits are either filed with this report or incorporated by reference into this report. See (b) Exhibits, which follow.
(b) Exhibits.
(2)Separation
| (2.1) | |||||
| (2.2) | |||||
| (2.3) | |||||
(3)Articles of Incorporation and bylaws
| (3.1) | |||||
| (3.2) | |||||
(4)Instruments defining the rights of security holders, including indentures
| (4.1) | |||||
| (4.2) | |||||
| (4.3) | |||||
(10)Material contracts and management compensation plans and arrangements:
| (10.1) | |||||
| (10.2) | |||||
| (10.3) | |||||
| (10.4) | |||||
105
| (10.5) | |||||
| (10.6) | |||||
| (10.7) | |||||
| (10.8) | |||||
| (10.9) | |||||
| (10.10) | |||||
| (10.11) | |||||
| (10.12) | |||||
| (10.13) | |||||
| (10.14) | |||||
| (10.15) | |||||
| (10.16) | |||||
| (10.17) | |||||
| (10.18) | |||||
| (10.19) | |||||
| (10.20) | |||||
| (10.21) | |||||
| (10.22) | |||||
| (10.23) | |||||
| (10.24) | |||||
| (10.25) | |||||
| (10.26) | |||||
| (10.27) | |||||
| (10.28) | |||||
| (10.29) | |||||
106
| (10.30) | |||||
| (10.31) | |||||
Filed herewith, unless otherwise indicated:
| (19) | |||||
| (21) | |||||
| (23) | |||||
| (31.1) | |||||
| (31.2) | |||||
| (32.1) | |||||
| (32.2) | |||||
(97) | |||||
| (101.INS) | Inline XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document) | ||||
| (101.SCH) | Inline XBRL Taxonomy Extension Schema Document | ||||
| (101.CAL) | Inline XBRL Taxonomy Extension Calculation Linkbase Document | ||||
| (101.DEF) | Inline XBRL Taxonomy Extension Definition Linkbase Document | ||||
| (101.LAB) | Inline XBRL Taxonomy Extension Label Linkbase Document | ||||
| (101.PRE) | Inline XBRL Taxonomy Extension Presentation Linkbase Document | ||||
| (104) | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | ||||
+Certain confidential information contained in this document, marked by [***], has been omitted because it is both (i) not material and (ii) would be competitively harmful if publicly disclosed.
*Schedules and exhibits omitted pursuant to Item 601(a)(5) of Regulation S-K. The Company agrees to furnish a supplemental copy of any omitted schedule to the Securities and Exchange Commission (the "SEC") upon request.
† Management contract or compensatory plan or arrangement.
Item 16. Form 10-K Summary
Registrants may voluntarily include a summary of information required by Form 10-K under this Item 16. The Company has elected to not include such summary information.
107
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
SOLVENTUM CORPORATION
(Registrant)
| Date: February 27, 2026 | |||||||||||
| By | /s/ Wayde McMillan | ||||||||||
| Wayde McMillan, | |||||||||||
| Executive Vice President and Chief Financial Officer (Mr. McMillan is the Principal Financial Officer and has been duly authorized to sign on behalf of the Registrant.) | |||||||||||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities indicated on February 27, 2026.
| Signature | Title | |||||||
| /s/ Bryan Hanson | Bryan Hanson, Chief Executive Officer (Principal Executive Officer and Director) | |||||||
| /s/ Wayde McMillan | Wayde McMillan, Executive Vice President, Chief Financial Officer (Principal Financial Officer) | |||||||
| /s/ Mary Wilcox | Mary Wilcox, Senior Vice President, Corporate Controller and Chief Accounting Officer (Principal Accounting Officer) | |||||||
/s/Carlos Albán | Carlos Albán, Director | |||||||
/s/Carrie S. Cox | Carrie S. Cox, Chair of the Board of Directors | |||||||
/s/Susan D. DeVore | Susan D. DeVore, Director | |||||||
/s/Shirley Edwards | Shirley Edwards, Director | |||||||
/s/Glenn A. Eisenberg | Glenn A. Eisenberg, Director | |||||||
/s/Dr. Bernard A. Harris Jr. | Dr. Bernard A. Harris Jr., Director | |||||||
/s/Karen J. May | Karen J. May, Director | |||||||
/s/Elizabeth A. Mily | Elizabeth A. Mily, Director | |||||||
/s/John H. Weiland | John H. Weiland, Director | |||||||
/s/Amy A. Wendell | Amy A. Wendell, Director | |||||||
/s/Darryl L. Wilson | Darryl L. Wilson, Director | |||||||
108